Pannexin 1 sustains the electrophysiological responsiveness of retinal ganglion cells
- PMID: 29643381
- PMCID: PMC5895610
- DOI: 10.1038/s41598-018-23894-2
Pannexin 1 sustains the electrophysiological responsiveness of retinal ganglion cells
Abstract
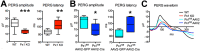
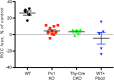
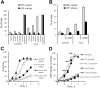
Pannexin 1 (Panx1) forms ATP-permeable membrane channels that play a key role in purinergic signaling in the nervous system in both normal and pathological conditions. In the retina, particularly high levels of Panx1 are found in retinal ganglion cells (RGCs), but the normal physiological function in these cells remains unclear. In this study, we used patch clamp recordings in the intact inner retina to show that evoked currents characteristic of Panx1 channel activity were detected only in RGCs, particularly in the OFF-type cells. The analysis of pattern electroretinogram (PERG) recordings indicated that Panx1 contributes to the electrical output of the retina. Consistently, PERG amplitudes were significantly impaired in the eyes with targeted ablation of the Panx1 gene in RGCs. Under ocular hypertension and ischemic conditions, however, high Panx1 activity permeated cell membranes and facilitated the selective loss of RGCs or stably transfected Neuro2A cells. Our results show that high expression of the Panx1 channel in RGCs is essential for visual function in the inner retina but makes these cells highly sensitive to mechanical and ischemic stresses. These findings are relevant to the pathophysiology of retinal disorders induced by increased intraocular pressure, such as glaucoma.
Conflict of interest statement
The authors declare no competing interests.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

