Calcium-sensing receptor (CaSR) promotes development of bone metastasis in renal cell carcinoma
- PMID: 29644008
- PMCID: PMC5884663
- DOI: 10.18632/oncotarget.24607
Calcium-sensing receptor (CaSR) promotes development of bone metastasis in renal cell carcinoma
Abstract
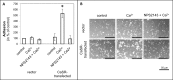
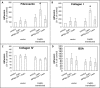
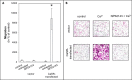
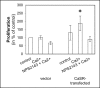
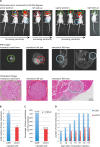
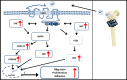
Bone metastasis is an important prognostic factor in renal cell carcinoma (RCC). The calcium-sensing receptor (CaSR) has been associated with bone metastasis in several different malignancies. We analyzed the impact of CaSR in bone metastasis in RCC in vitro and in vivo. The RCC cell line 786-O was stably transfected with the CaSR gene and treated with calcium alone or in combination with the CaSR antagonist NPS2143. Afterwards migration, adhesion, proliferation and prominent signaling molecules were analyzed. Calcium treated CaSR-transfected 768-O cells showed an increased adhesion to endothelial cells and the extracellular matrix components fibronectin and collagen I, but not to collagen IV. The chemotactic cell migration and proliferation was also induced by calcium. The activity of SHC, AKT, ERK, P90RSK and JNK were enhanced after calcium treatment of CaSR-transfected cells. These effects were abolished by NPS2143. Development of bone metastasis was evaluated in vivo in a mouse model. Intracardiac injection of CaSR-transfected 768-O cells showed an increased rate of bone metastasis. The results indicate CaSR as an important component in the mechanism of bone metastasis in RCC. Therefore, targeting CaSR might be beneficial in patients with bone metastatic RCC with a high CaSR expression.
Keywords: bone metastases, metastasis; calcium-sensing receptor; kidney cancer; renal cell carcinoma.
Conflict of interest statement
CONFLICTS OF INTEREST No conflict of interest.
Figures

References
-
- Society AC . Cancer Facts & Figures 2016. Atlanta: American Cancer Society; 2016.
-
- Chow WH, Devesa SS, Warren JL, Fraumeni JF., Jr Rising incidence of renal cell cancer in the United States. JAMA. 1999;281:1628–31. - PubMed
-
- Decastro GJ, McKiernan JM. Epidemiology, clinical staging, and presentation of renal cell carcinoma. Urol Clin North Am. 2008;35:581–92. https://doi.org/10.1016/j.ucl.2008.07.005. - DOI - PubMed
-
- Sun M, Choueiri TK. Kidney cancer: Recurrence in renal cell carcinoma: the work is not done. Nat Rev Urol. 2016;13:246–7. https://doi.org/10.1038/nrurol.2016.57. - DOI - PubMed
-
- Mikami S, Oya M, Mizuno R, Kosaka T, Katsube KI, Okada Y. Invasion and metastasis of renal cell carcinoma. Medical molecular morphology. 2014;47:63–7. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

