Synthesis and Application of the Transition Metal Complexes of α-Pyridinyl Alcohols, α-Bipyridinyl Alcohols, α,α'-Pyridinyl Diols and α,α'-Bipyridinyl Diols in Homogeneous Catalysis
- PMID: 29649178
- PMCID: PMC6017785
- DOI: 10.3390/molecules23040896
Synthesis and Application of the Transition Metal Complexes of α-Pyridinyl Alcohols, α-Bipyridinyl Alcohols, α,α'-Pyridinyl Diols and α,α'-Bipyridinyl Diols in Homogeneous Catalysis
Abstract
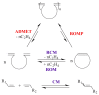
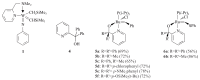
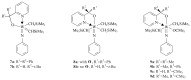
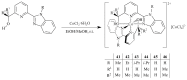
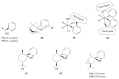
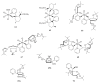
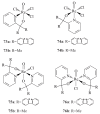
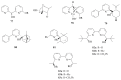
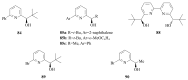
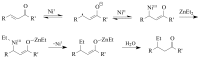
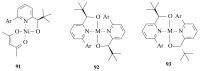
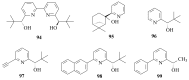
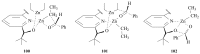
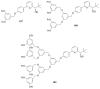
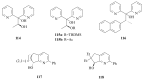
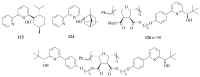
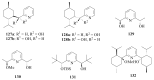
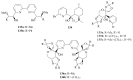
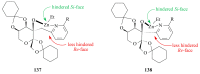
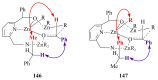
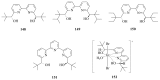
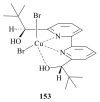
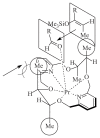
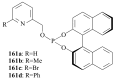
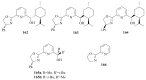
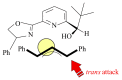
The paper presents a comprehensive survey on the synthetic procedures of transition metal complexes of α-pyridinyl alcoholato, α-bipyridinyl alcoholato, α,α'-pyridinyl dialcoholato and α,α'-bipyridinyl dialcoholato ligands and their coordination chemistry. Greater emphasis is, however, given to the catalytic activity of the complexes in homogeneous and asymmetric chemical reactions. The multidentate character of the pyridinyl alcohols and/or bipyridinyl diols is of great importance in the complexation with a large number and type of transition metals. The transition metal complexes of pyridinyl alcoholato or bipyridinyl dialcoholato ligands in most cases, and a few pyridinyl alcohols alone, were used as catalysts in homogeneous and chemical asymmetric reactions. In most of the homogeneously catalysed enantioselective chemical reactions, limited numbers and types of pyridinyl alcohols and or bipyridinyl diols were used in the preparation of chiral catalysts that led to a few investigations on the catalytic importance of the pyridinyl alcohols.
Keywords: bipyridinyl diol; catalyst; enantioselective; pyridinyl alcohol; synthesis; transition metal complex.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
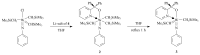



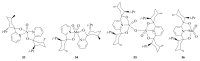
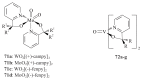
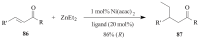
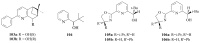

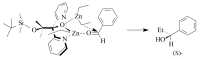

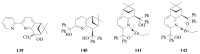
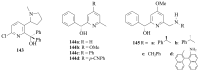
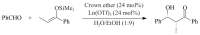


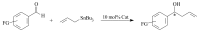



Similar articles
-
α-Pyridinyl Alcohols, α,α'-Pyridine Diols, α-Bipyridinyl Alcohols, and α,α'-Bipyridine Diols as Structure Motifs Towards Important Organic Molecules and Transition Metal Complexes.Curr Org Synth. 2020;17(5):344-366. doi: 10.2174/1570179417666200212111049. Curr Org Synth. 2020. PMID: 32091340
-
ProPhenol-catalyzed asymmetric additions by spontaneously assembled dinuclear main group metal complexes.Acc Chem Res. 2015 Mar 17;48(3):688-701. doi: 10.1021/ar500374r. Epub 2015 Feb 4. Acc Chem Res. 2015. PMID: 25650587 Free PMC article.
-
N-Alkylation by Hydrogen Autotransfer Reactions [corrected].Top Curr Chem (Cham). 2016 Jun;374(3):27. doi: 10.1007/s41061-016-0027-1. Epub 2016 Apr 29. Top Curr Chem (Cham). 2016. PMID: 27573267
-
Advances in Chiral Pincer Complexes: Insights and Applications in Catalytic Asymmetric Reactions.Int J Mol Sci. 2024 Sep 26;25(19):10344. doi: 10.3390/ijms251910344. Int J Mol Sci. 2024. PMID: 39408673 Free PMC article. Review.
-
Recent advances in osmium-catalyzed hydrogenation and dehydrogenation reactions.Acc Chem Res. 2015 Feb 17;48(2):363-79. doi: 10.1021/ar5003818. Epub 2015 Feb 4. Acc Chem Res. 2015. PMID: 25650714 Review.
Cited by
-
Catalysis of linear alkene metathesis by Grubbs-type ruthenium alkylidene complexes containing hemilabile α,α-diphenyl-(monosubstituted-pyridin-2-yl)methanolato ligands.Beilstein J Org Chem. 2019 Jan 22;15:194-209. doi: 10.3762/bjoc.15.19. eCollection 2019. Beilstein J Org Chem. 2019. PMID: 30745994 Free PMC article.
-
Soft synthesis and characterization of goethite-based nanocomposites as promising cyclooctene oxidation catalysts.RSC Adv. 2021 Aug 13;11(44):27589-27602. doi: 10.1039/d1ra04211d. eCollection 2021 Aug 9. RSC Adv. 2021. PMID: 35480697 Free PMC article.
References
-
- Hagen J. Industrial Catalysis: A Practical Approach. 2nd ed. Wiley-VCH Verlag; Weinheim, Germany: 2006.
-
- Fletcher N.C. Chiral 2,2′-bipyridines: Ligands for asymmetric induction. J. Chem. Soc. Perkin Trans. 2002;1:1831–1842. doi: 10.1039/B204272J. - DOI
-
- Pfaltz A. In: Enantioselective Catalysis with Chiral Co- and Cu-Complexes. Scheffold R., editor. Volume 5. Springer; Berlin, Germany: 1989. pp. 199–248. Modern Synthetic Methods.
-
- Hegetschweiler K., Schmalle H., Streit H.M., Schneider W. Synthesis and structure of a novel hexanuclear iron (III) complex containing six terminal and twelve bridging groups and one μ6-oxo bridge. Inorg. Chem. 1990;29:3625–3627. doi: 10.1021/ic00343a066. - DOI
-
- Soai K., Niwa S. Enantioselective addition of organozinc reagents to aldehydes. Chem. Rev. 1992;92:833–856. doi: 10.1021/cr00013a004. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

