Selective inhibition of intestinal guanosine 3',5'-cyclic monophosphate signaling by small-molecule protein kinase inhibitors
- PMID: 29653944
- PMCID: PMC5971447
- DOI: 10.1074/jbc.RA118.002835
Selective inhibition of intestinal guanosine 3',5'-cyclic monophosphate signaling by small-molecule protein kinase inhibitors
Abstract
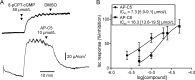
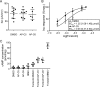
The guanosine 3',5'-cyclic monophosphate (cGMP)-dependent protein kinase II (cGKII) serine/threonine kinase relays signaling through guanylyl cyclase C (GCC) to control intestinal fluid homeostasis. Here, we report the discovery of small-molecule inhibitors of cGKII. These inhibitors were imidazole-aminopyrimidines, which blocked recombinant human cGKII at submicromolar concentrations but exhibited comparatively little activity toward the phylogenetically related protein kinases cGKI and cAMP-dependent protein kinase (PKA). Whereas aminopyrimidyl motifs are common in protein kinase inhibitors, molecular modeling of these imidazole-aminopyrimidines in the ATP-binding pocket of cGKII indicated an unconventional binding mode that directs their amine substituent into a narrow pocket delineated by hydrophobic residues of the hinge and the αC-helix. Crucially, this set of residues included the Leu-530 gatekeeper, which is not conserved in cGKI and PKA. In intestinal organoids, these compounds blocked cGKII-dependent phosphorylation of the vasodilator-stimulated phosphoprotein (VASP). In mouse small intestinal tissue, cGKII inhibition significantly attenuated the anion secretory response provoked by the GCC-activating bacterial heat-stable toxin (STa), a frequent cause of infectious secretory diarrhea. In contrast, both PKA-dependent VASP phosphorylation and intestinal anion secretion were unaffected by treatment with these compounds, whereas experiments with T84 cells indicated that they weakly inhibit the activity of cAMP-hydrolyzing phosphodiesterases. As these protein kinase inhibitors are the first to display selective inhibition of cGKII, they may expedite research on cGMP signaling and may aid future development of therapeutics for managing diarrheal disease and other pathogenic syndromes that involve cGKII.
Keywords: CFTR; ETEC; PKG; cGMP; cyclic GMP; cystic fibrosis transmembrane conductance regulator; drug discovery; enterotoxigenic E. coli; intestinal epithelium; protein kinase G; secretory diarrhea; signal transduction; vasodilator-stimulated phosphoprotein.
© 2018 Bijvelds et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures





References
-
- de Jonge H. R. (1981) Cyclic GMP-dependent protein kinase in intestinal brush borders. Adv. Cyclic Nucleotide Res. 14, 315–333 - PubMed
-
- Jarchau T., Häusler C., Markert T., Pöhler D., Vanderkerckhove J., De Jonge H. R., Lohmann S. M., and Walter U. (1994) Cloning, expression, and in situ localization of rat intestinal cGMP-dependent protein kinase II. Proc. Natl. Acad. Sci. U.S.A. 91, 9426–9430 10.1073/pnas.91.20.9426 - DOI - PMC - PubMed
-
- el-Husseini A. E., Bladen C., and Vincent S. R. (1995) Molecular characterization of a type II cyclic GMP-dependent protein kinase expressed in the rat brain. J. Neurochem. 64, 2814–2817 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

