Efficacy, Safety, and Potency of Betamethasone Dipropionate Spray 0.05%: A Treatment for Adults with Mild-to-moderate Plaque Psoriasis
- PMID: 29657667
- PMCID: PMC5891082
Efficacy, Safety, and Potency of Betamethasone Dipropionate Spray 0.05%: A Treatment for Adults with Mild-to-moderate Plaque Psoriasis
Abstract
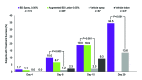
Objective: A spray formulation of betamethasone dipropionate 0.05% (BD spray 0.05%; Sernivo™ [betamethasone dipropionate] Spray 0.05%; Promius Pharma, LLC; Princeton, New Jersey, USA) has been developed for the topical treatment of psoriasis. The objective of these studies was to evaluate the efficacy, safety, and potency of BD spray 0.05%. Design, Setting, Participants, and Measurements: Efficacy and safety were assessed in a randomized, vehicle-controlled, double-blind study in adults with moderate plaque psoriasis (ClinicalTrials.gov identifier: NCT01947491). Additionally, the potential for adrenal suppression and systemic absorption was evaluated in a randomized, open-label study in healthy adults (ClinicalTrials.gov identifier: NCT02070965). Potency was measured in two single-point, randomized, evaluator-blinded studies in healthy adults. Results: BD spray 0.05% was significantly more effective than the vehicle spray in subjects with moderate plaque psoriasis after three, 14, and 28 days of twice-daily treatment. The efficacy of BD spray 0.05% was similar to augmented BD lotion 0.05% after 14 days of treatment. The safety of BD spray 0.05% was similar to that of the vehicle spray over 28 days and to that of augmented BD lotion 0.05% over 14 days. Under maximal use conditions for up to 29 days, the potential for adrenal suppression was no greater with BD spray 0.05% than with a 15-day regimen of augmented BD lotion 0.05%. There was less systemic absorption of BD from BD spray 0.05% than from augmented BD lotion 0.05%. Studies classify BD spray 0.05% as a midpotent corticosteroid. Conclusions: BD spray 0.05%, a midpotent corticosteroid, is an effective and well-tolerated treatment for adults with mild to moderate plaque psoriasis.
Keywords: Psoriasis; betamethasone dipropionate; corticosteroid; topical.
Conflict of interest statement
FUNDING:This study was funded and sponsored by the Dr. Reddy’s Laboratories group of companies in Princeton, New Jersey, USA DISCLOSURES:Drs. Sidgiddi, Pakunlu, and Allenby are employees of Dr. Reddy’s Laboratories, whose subsidiary, Promius Pharma, LLC, markets the topical formulation of betamethasone dipropionate spray, 0.05% (Sernivo™), the product used in this study. Drs. Sidgiddi and Allenby own stock in Dr. Reddy’s Laboratories.
Figures

Similar articles
-
DFD-01, a Novel Medium Potency Betamethasone Dipropionate 0.05% Emollient Spray, Demonstrates Similar Efficacy to Augmented Betamethasone Dipropionate 0.05% Lotion for the Treatment of Moderate Plaque Psoriasis.J Drugs Dermatol. 2016 Feb;15(2):154-62. J Drugs Dermatol. 2016. PMID: 26885782 Clinical Trial.
-
DFD-01: a novel topical formulation of betamethasone dipropionate for the treatment of extensive psoriasis.Expert Rev Clin Immunol. 2017 Feb;13(2):85-91. doi: 10.1080/1744666X.2017.1270756. Epub 2016 Dec 21. Expert Rev Clin Immunol. 2017. PMID: 27967262 Review.
-
Betamethasone dipropionate spray 0.05% alleviates troublesome symptoms of plaque psoriasis.Cutis. 2020 Feb;105(2):97-102;E1. Cutis. 2020. PMID: 32186532
-
Efficacy and safety of twice-daily augmented betamethasone dipropionate lotion versus clobetasol propionate solution in patients with moderate-to-severe scalp psoriasis.Clin Ther. 1995 May-Jun;17(3):390-401. doi: 10.1016/0149-2918(95)80104-9. Clin Ther. 1995. PMID: 7585843 Clinical Trial.
-
Calcipotriene and betamethasone dipropionate for the topical treatment of plaque psoriasis.Expert Rev Clin Pharmacol. 2016 Jun;9(6):789-97. doi: 10.1080/17512433.2016.1179574. Epub 2016 May 13. Expert Rev Clin Pharmacol. 2016. PMID: 27089906 Review.
References
-
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70(3):512–516. - PubMed
-
- National Psoriasis Foundation. [Accessed July 12, 2017]. https://www.psoriasis.org/sites/default/files/psoriasis_fact_sheet.pdf About psoriasis. Available at.
-
- Menter A, Korman NJ, Elmets CA, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis. Section 3. Guidelines of care for the management and treatment of psoriasis with topical therapies. J Am Acad Dermatol. 2009;60(4):643–659. - PubMed
-
- Nordwall C. Local treatment of psoriasis and eczema with betamethasone-17,21-dipropionate (Diproderm cream): a double-blind comparsion with fluocortolone caproate, fluocortolone pivalate (Ultralanum cream) Curr Ther Res Clin Exp. 1974;16(8):798–803. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical
