Optimised ARID1A immunohistochemistry is an accurate predictor of ARID1A mutational status in gynaecological cancers
- PMID: 29659191
- PMCID: PMC6065117
- DOI: 10.1002/cjp2.103
Optimised ARID1A immunohistochemistry is an accurate predictor of ARID1A mutational status in gynaecological cancers
Abstract
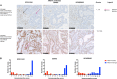
ARID1A is a tumour suppressor gene that is frequently mutated in clear cell and endometrioid carcinomas of the ovary and endometrium and is an important clinical biomarker for novel treatment approaches for patients with ARID1A defects. However, the accuracy of ARID1A immunohistochemistry (IHC) as a surrogate for mutation status has not fully been established for patient stratification in clinical trials. Here we tested whether ARID1A IHC could reliably predict ARID1A mutations identified by next-generation sequencing. Three commercially available antibodies - EPR13501 (Abcam), D2A8U (Cell Signaling), and HPA005456 (Sigma) - were optimised for IHC using cell line models and human tissue, and screened across a cohort of 45 gynaecological tumours. IHC was scored independently by three pathologists using an immunoreactive score. ARID1A mutation status was assessed using two independent sequencing platforms and the concordance between ARID1A mutation and protein expression was evaluated using Receiver Operating Characteristic statistics. Overall, 21 ARID1A mutations were identified in 14/43 assessable tumours (33%), the majority of which were predicted to be deleterious. Mutations were identified in 6/17 (35%) ovarian clear cell carcinomas, 5/8 (63%) ovarian endometrioid carcinomas, 2/5 (40%) endometrial carcinomas, and 1/7 (14%) carcinosarcomas. ROC analysis identified greater than 95% concordance between mutation status and IHC using a modified immunoreactive score for all three antibodies allowing a definitive cut-point for ARID1A mutant status to be calculated. Comprehensive assessment of concordance of ARID1A IHC and mutation status identified EPR13501 as an optimal antibody, with 100% concordance between ARID1A mutation status and protein expression, across different gynaecological histological subtypes. It delivered the best inter-rater agreement between all pathologists, as well as a clear cost-benefit advantage. This could allow patients to be accurately stratified based on their ARID1A IHC status into early phase clinical trials.
Keywords: ARID1A immunohistochemistry; biomarker; clear cell ovarian cancer; next generation sequencing.
© 2018 The Authors The Journal of Pathology: Clinical Research published by The Pathological Society of Great Britain and Ireland and John Wiley & Sons Ltd.
Figures




Comment in
-
You won't believe this old test … that does cheap single-cell mutation detection.J Pathol Clin Res. 2018 Jul;4(3):149-153. doi: 10.1002/cjp2.108. J Pathol Clin Res. 2018. PMID: 30003713 Free PMC article.
References
-
- Hembree TN, Teer JK, Hakam A, et al Genetic investigation of uterine carcinosarcoma: case report and cohort analysis. Cancer Control 2016; 23 : 61–66. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

