Development of Macrocyclic Peptidomimetics Containing Constrained α,α-Dialkylated Amino Acids with Potent and Selective Activity at Human Melanocortin Receptors
- PMID: 29660981
- PMCID: PMC5999404
- DOI: 10.1021/acs.jmedchem.8b00488
Development of Macrocyclic Peptidomimetics Containing Constrained α,α-Dialkylated Amino Acids with Potent and Selective Activity at Human Melanocortin Receptors
Abstract
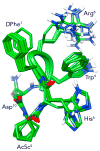
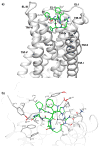
We report the development of macrocyclic melanocortin derivatives of MT-II and SHU-9119, achieved by modifying the cycle dimension and incorporating constrained amino acids in ring-closing. This study culminated in the discovery of novel agonists/antagonists with an unprecedented activity profile by adding pieces to the puzzle of the melanocortin receptor selectivity. Finally, the resulting 19- and 20-membered rings represent a suitable frame for the design of further therapeutic ligands as selective modulators of the melanocortin system.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
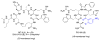

Similar articles
-
Design of novel melanotropin agonists and antagonists with high potency and selectivity for human melanocortin receptors.Peptides. 2005 Aug;26(8):1481-5. doi: 10.1016/j.peptides.2005.03.020. Peptides. 2005. PMID: 15876475 Review.
-
When Macrocyclic Peptides Meet the Crystal Structure of a Melanocortin Receptor.J Med Chem. 2021 Jan 14;64(1):354-356. doi: 10.1021/acs.jmedchem.0c02087. Epub 2021 Jan 4. J Med Chem. 2021. PMID: 33393773 Free PMC article.
-
Discovery of Novel Potent and Selective Agonists at the Melanocortin-3 Receptor.J Med Chem. 2015 Dec 24;58(24):9773-8. doi: 10.1021/acs.jmedchem.5b01285. Epub 2015 Dec 16. J Med Chem. 2015. PMID: 26599352 Free PMC article.
-
New tripeptide-based macrocyclic calpain inhibitors formed by N-alkylation of histidine.Chem Biodivers. 2012 Nov;9(11):2473-84. doi: 10.1002/cbdv.201200320. Chem Biodivers. 2012. PMID: 23161629
-
Design of novel melanocortin receptor ligands: multiple receptors, complex pharmacology, the challenge.Eur J Pharmacol. 2011 Jun 11;660(1):88-93. doi: 10.1016/j.ejphar.2010.10.109. Epub 2011 Jan 3. Eur J Pharmacol. 2011. PMID: 21208601 Free PMC article. Review.
Cited by
-
Demonstration of a Common DPhe7 to DNal(2')7 Peptide Ligand Antagonist Switch for Melanocortin-3 and Melanocortin-4 Receptors Identifies the Systematic Mischaracterization of the Pharmacological Properties of Melanocortin Peptides.J Med Chem. 2022 Apr 28;65(8):5990-6000. doi: 10.1021/acs.jmedchem.1c01295. Epub 2022 Apr 11. J Med Chem. 2022. PMID: 35404053 Free PMC article.
-
Ocular pharmacological and biochemical profiles of 6-thioguanine: a drug repurposing study.Front Pharmacol. 2024 Mar 25;15:1375805. doi: 10.3389/fphar.2024.1375805. eCollection 2024. Front Pharmacol. 2024. PMID: 38590636 Free PMC article.
-
Breast Tumor Cell Invasion and Pro-Invasive Activity of Cancer-Associated Fibroblasts Co-Targeted by Novel Urokinase-Derived Decapeptides.Cancers (Basel). 2020 Aug 24;12(9):2404. doi: 10.3390/cancers12092404. Cancers (Basel). 2020. PMID: 32847144 Free PMC article.
-
Fingolimod and Diabetic Retinopathy: A Drug Repurposing Study.Front Pharmacol. 2021 Sep 17;12:718902. doi: 10.3389/fphar.2021.718902. eCollection 2021. Front Pharmacol. 2021. PMID: 34603029 Free PMC article.
-
Recommended Tool Compounds for the Melanocortin Receptor (MCR) G Protein-Coupled Receptors (GPCRs).ACS Pharmacol Transl Sci. 2024 Aug 26;7(9):2706-2724. doi: 10.1021/acsptsci.4c00129. eCollection 2024 Sep 13. ACS Pharmacol Transl Sci. 2024. PMID: 39296259 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information

