A simplified scoring system in de novo follicular lymphoma treated initially with immunochemotherapy
- PMID: 29666118
- PMCID: PMC6034646
- DOI: 10.1182/blood-2017-11-816405
A simplified scoring system in de novo follicular lymphoma treated initially with immunochemotherapy
Abstract
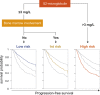
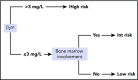
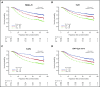
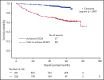
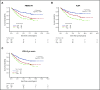
In follicular lymphoma (FL), no prognostic index has been built based solely on a cohort of patients treated with initial immunochemotherapy. There is currently a need to define parsimonious clinical models for trial stratification and to add on biomolecular factors. Here, we confirmed the validity of both the follicular lymphoma international prognostic index (FLIPI) and the FLIPI2 in the large prospective PRIMA trial cohort of 1135 patients treated with initial R-chemotherapy ± R maintenance. Furthermore, we developed a new prognostic tool comprising only 2 simple parameters (bone marrow involvement and β2-microglobulin [β2m]) to predict progression-free survival (PFS). The final simplified score, called the PRIMA-PI (PRIMA-prognostic index), comprised 3 risk categories: high (β2m > 3 mg/L), low (β2m ≤ 3 mg/L without bone marrow involvement), and intermediate (β2m ≤ 3 mg/L with bone marrow involvement). Five-year PFS rates were 69%, 55%, and 37% in the low-, intermediate-, and high-risk groups, respectively (P < .0001). In addition, achieving event-free survival (EFS) or not at 24 months (EFS24) was a strong posttreatment prognostic parameter for subsequent overall survival, and the PRIMA-PI was correlated with EFS24. The results were confirmed in a pooled external validation cohort of 479 patients from the FL2000 LYSA trial and the University of Iowa/Mayo Clinic Lymphoma Specialized Program of Research Excellence Molecular Epidemiology Resource. Five-year EFS in the validation cohort was 77%, 57%, and 44% in the PRIMA-PI low-, intermediate-, and high-risk groups, respectively (P < .0001). The PRIMA-PI is a novel and easy-to-compute prognostic index for patients initially treated with immunochemotherapy. This could serve as a basis for building more sophisticated and integrated biomolecular scores.
© 2018 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Figures
Comment in
-
The FLIPI nowadays: validation and modification.Blood. 2018 Jul 5;132(1):3-4. doi: 10.1182/blood-2018-05-847558. Blood. 2018. PMID: 29976777 No abstract available.
References
-
- Teras LR, DeSantis CE, Cerhan JR, Morton LM, Jemal A, Flowers CR. 2016 US lymphoid malignancy statistics by World Health Organization subtypes [published online ahead of print 12 September 2016]. CA Cancer J Clin. doi:10.3322/caac.21357. - PubMed
-
- Link BK, Maurer MJ, Nowakowski GS, et al. Rates and outcomes of follicular lymphoma transformation in the immunochemotherapy era: a report from the University of Iowa/MayoClinic Specialized Program of Research Excellence Molecular Epidemiology Resource. J Clin Oncol. 2013;31(26):3272-3278. - PMC - PubMed
-
- Brice P, Bastion Y, Lepage E, et al. Comparison in low-tumor-burden follicular lymphomas between an initial no-treatment policy, prednimustine, or interferon alfa: a randomized study from the Groupe d’Etude des Lymphomes Folliculaires. Groupe d’Etude des Lymphomes de l’Adulte. J Clin Oncol. 1997;15(3):1110-1117. - PubMed
-
- Herold M, Haas A, Srock S, et al. ; East German Study Group Hematology and Oncology Study. Rituximab added to first-line mitoxantrone, chlorambucil, and prednisolone chemotherapy followed by interferon maintenance prolongs survival in patients with advanced follicular lymphoma: an East German Study Group Hematology and Oncology Study. J Clin Oncol. 2007;25(15):1986-1992. - PubMed
-
- Hiddemann W, Kneba M, Dreyling M, et al. Frontline therapy with rituximab added to the combination of cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) significantly improves the outcome for patients with advanced-stage follicular lymphoma compared with therapy with CHOP alone: results of a prospective randomized study of the German Low-Grade Lymphoma Study Group. Blood. 2005;106(12):3725-3732. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

