Pharmacokinetic equivalence study of nonsteroidal anti-inflammatory drug etoricoxib
- PMID: 29670410
- PMCID: PMC5896653
- DOI: 10.2147/CPAA.S161024
Pharmacokinetic equivalence study of nonsteroidal anti-inflammatory drug etoricoxib
Abstract
Purpose: The current study aimed to evaluate whether a generic product of etoricoxib 120 mg film-coated tablet (the test drug) was bioequivalent to the reference product (Arcoxia® film-coated tablet 120 mg).
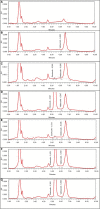
Methods: This was a randomized, open-label, two-sequence, crossover study under fasting condition, with a 14-day washout period, involving 26 healthy adult male and female subjects. Blood samples were taken and analyzed for plasma concentrations of etoricoxib (Chemical Abstracts Service [CAS] 202409-33-4) using a high-pressure liquid chromatography-ultraviolet detector (HPLC-UV) system capable of measuring etoricoxib concentrations ranging from 5.00 to 5002.90 ng/mL, with the lowest limit of quantitation of 5.00 ng/mL. A noncompartmental method was used to determine the pharmacokinetic parameters of a single-dose administration of the drug, including the area under plasma concentration-time curve from time zero to the time of last observed concentration (AUC0-t ), the area under plasma concentration-time curve from time zero to infinity (AUC0-∞), the maximum plasma concentration (Cmax), the time to reach the maximum plasma concentration (tmax), and the terminal half-life (t½).
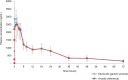
Results: After a single-dose administration of etoricoxib 120 mg film-coated tablet, the mean (SD) values for the AUC0-72h and Cmax of the test drug were 45913.42 (13142.19) ng·h/mL and 3155.93 (752.81) ng/mL, respectively; the values for the reference drug were 44577.20 (13541.85) ng·h/mL and 2915.13 (772.81) ng/mL, respectively. The geometric mean ratios (90% CIs) of the test drug/reference drug were 103.40% (98.70%-108.32%) for AUC0-72h and 109.26% (100.18%-119.18%) for Cmax. No clinically significant differences in tmax and t½values were found between the test drug and the reference drug. No adverse events were experienced by the subjects during this study.
Conclusion: The present study demonstrated that the evaluated generic etoricoxib 120 mg film-coated tablets were bioequivalent to the reference drug.
Keywords: bioavailability; bioequivalence; etoricoxib; nonsteroidal anti-inflammatory drug; selective cyclooxygenase-2 inhibitor.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures
Similar articles
-
Pharmacokinetic equivalence study of two formulations of the anticonvulsant pregabalin.Clin Pharmacol. 2015 Apr 20;7:69-75. doi: 10.2147/CPAA.S82143. eCollection 2015. Clin Pharmacol. 2015. PMID: 25945069 Free PMC article.
-
Pharmacokinetic and bioequivalence study of etoricoxib tablet in healthy Bangladeshi volunteers.Arzneimittelforschung. 2011;61(11):617-21. doi: 10.1055/s-0031-1300564. Arzneimittelforschung. 2011. PMID: 22232850 Clinical Trial.
-
Pharmacokinetics and bioequivalence of Ezetimibe tablet versus Ezetrol®:an open-label, randomized, two-sequence crossover study in healthy Chinese subjects.BMC Pharmacol Toxicol. 2023 Feb 3;24(1):7. doi: 10.1186/s40360-023-00649-y. BMC Pharmacol Toxicol. 2023. PMID: 36737825 Free PMC article. Clinical Trial.
-
Etoricoxib Coated Tablets: Bioequivalence Assessment between Two Formulations Administered under Fasting Conditions.Pharmaceutics. 2023 Nov 1;15(11):2569. doi: 10.3390/pharmaceutics15112569. Pharmaceutics. 2023. PMID: 38004548 Free PMC article.
-
Bioavailability of omeprazole 20 mg capsules containing omeprazole 22.5% enteric coated pellets versus a reference product in healthy Bangladeshi male subjects: an open-label, single-dose, randomized-sequence, two-way crossover study.Int J Clin Pharmacol Ther. 2011 Dec;49(12):778-86. doi: 10.5414/cp201569. Int J Clin Pharmacol Ther. 2011. PMID: 22122821 Clinical Trial.
Cited by
-
Merging konjac glucomannan with other copolymeric hydrogels as a cutting-edge liquid raft system for dual delivery of etoricoxib and famotidine.Drug Deliv. 2023 Dec;30(1):2189630. doi: 10.1080/10717544.2023.2189630. Drug Deliv. 2023. PMID: 36927148 Free PMC article.
-
Fast and Sensitive HPLC-ESI-MS/MS Method for Etoricoxib Quantification in Human Plasma and Application to Bioequivalence Study.Molecules. 2022 Sep 4;27(17):5706. doi: 10.3390/molecules27175706. Molecules. 2022. PMID: 36080473 Free PMC article.
-
A Phase I, open-label, randomized, crossover study in healthy subjects to evaluate the bioavailability of, and the food effect on, a pomalidomide oral liquid suspension.Clin Pharmacol. 2018 Jul 19;10:89-99. doi: 10.2147/CPAA.S171735. eCollection 2018. Clin Pharmacol. 2018. PMID: 30050331 Free PMC article.
-
Nanoformulations of Ursolic Acid: A Modern Natural Anticancer Molecule.Front Pharmacol. 2021 Jul 5;12:706121. doi: 10.3389/fphar.2021.706121. eCollection 2021. Front Pharmacol. 2021. PMID: 34295253 Free PMC article. Review.
-
Vitamin B and Vitamin C Affect DNA Methylation and Amino Acid Metabolism in Mycobacterium bovis BCG.Front Microbiol. 2020 Apr 22;11:812. doi: 10.3389/fmicb.2020.00812. eCollection 2020. Front Microbiol. 2020. PMID: 32390998 Free PMC article.
References
-
- WHO [webpage on the Internet] World Health Organization Essential Medicines and Health Products. 2017. [Accessed December 18, 2017]. Available from: www.who.int/medicines/services/essmedicines_def/en/
-
- FDA Facts about Generic Drugs. 2016. [Accessed December 18, 2017]. Available from: www.fda.gov/downloads/drugs/resourcesforyou/consumers/buyingusingmedicin....
-
- EMA [webpage on the Internet] Generic and Hybrid Medicines. 2012. [Accessed December 18, 2017]. Available from: www.ema.europa.eu/ema/index.jsp?curl=pages/special_topics/document_listi....
-
- FDA [webpage on the Internet] Generic Drugs. 2017. [Accessed December 18, 2017]. Available from: https://www.fda.gov/drugs/resourcesforyou/consumers/buyingusingmedicines....
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

