SALM/Lrfn Family Synaptic Adhesion Molecules
- PMID: 29674953
- PMCID: PMC5895706
- DOI: 10.3389/fnmol.2018.00105
SALM/Lrfn Family Synaptic Adhesion Molecules
Abstract
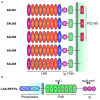
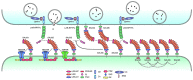
Synaptic adhesion-like molecules (SALMs) are a family of cell adhesion molecules involved in regulating neuronal and synapse development that have also been implicated in diverse brain dysfunctions, including autism spectrum disorders (ASDs). SALMs, also known as leucine-rich repeat (LRR) and fibronectin III domain-containing (LRFN) proteins, were originally identified as a group of novel adhesion-like molecules that contain LRRs in the extracellular region as well as a PDZ domain-binding tail that couples to PSD-95, an abundant excitatory postsynaptic scaffolding protein. While studies over the last decade have steadily explored the basic properties and synaptic and neuronal functions of SALMs, a number of recent studies have provided novel insights into molecular, structural, functional and clinical aspects of SALMs. Here we summarize these findings and discuss how SALMs act in concert with other synaptic proteins to regulate synapse development and function.
Keywords: Lrfn; PSD-95; SALM; adhesion molecules; synaptic.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

