CD81 Receptor Regions outside the Large Extracellular Loop Determine Hepatitis C Virus Entry into Hepatoma Cells
- PMID: 29677132
- PMCID: PMC5923501
- DOI: 10.3390/v10040207
CD81 Receptor Regions outside the Large Extracellular Loop Determine Hepatitis C Virus Entry into Hepatoma Cells
Abstract
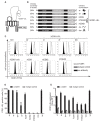
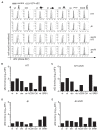
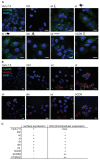
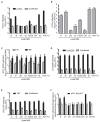
Hepatitis C virus (HCV) enters human hepatocytes using four essential entry factors, one of which is human CD81 (hCD81). The tetraspanin hCD81 contains a large extracellular loop (LEL), which interacts with the E2 glycoprotein of HCV. The role of the non-LEL regions of hCD81 (intracellular tails, four transmembrane domains, small extracellular loop and intracellular loop) is poorly understood. Here, we studied the contribution of these domains to HCV susceptibility of hepatoma cells by generating chimeras of related tetraspanins with the hCD81 LEL. Our results show that non-LEL regions in addition to the LEL determine susceptibility of cells to HCV. While closely related tetraspanins (X. tropicalis CD81 and D. rerio CD81) functionally complement hCD81 non-LEL regions, distantly related tetraspanins (C. elegans TSP9 amd D. melanogaster TSP96F) do not and tetraspanins with intermediate homology (hCD9) show an intermediate phenotype. Tetraspanin homology and susceptibility to HCV correlate positively. For some chimeras, infectivity correlates with surface expression. In contrast, the hCD9 chimera is fully surface expressed, binds HCV E2 glycoprotein but is impaired in HCV receptor function. We demonstrate that a cholesterol-coordinating glutamate residue in CD81, which hCD9 lacks, promotes HCV infection. This work highlights the hCD81 non-LEL regions as additional HCV susceptibility-determining factors.
Keywords: CD81; HCV; chimeras; cholesterol-binding residue; hepatitis C virus; receptor; susceptibility-determining domains; tetraspanin; transmembrane domain four.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

