In Vitro Identification of New Transcriptomic and miRNomic Profiles Associated with Pulmonary Fibrosis Induced by High Doses Everolimus: Looking for New Pathogenetic Markers and Therapeutic Targets
- PMID: 29677166
- PMCID: PMC5979287
- DOI: 10.3390/ijms19041250
In Vitro Identification of New Transcriptomic and miRNomic Profiles Associated with Pulmonary Fibrosis Induced by High Doses Everolimus: Looking for New Pathogenetic Markers and Therapeutic Targets
Abstract
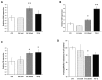
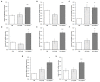
The administration of Everolimus (EVE), a mTOR inhibitor used in transplantation and cancer, is often associated with adverse effects including pulmonary fibrosis. Although the underlying mechanism is not fully clarified, this condition could be in part caused by epithelial to mesenchymal transition (EMT) of airway cells. To improve our knowledge, primary bronchial epithelial cells (BE63/3) were treated with EVE (5 and 100 nM) for 24 h. EMT markers (α-SMA, vimentin, fibronectin) were measured by RT-PCR. Transepithelial resistance was measured by Millicell-ERS ohmmeter. mRNA and microRNA profiling were performed by Illumina and Agilent kit, respectively. Only high dose EVE increased EMT markers and reduced the transepithelial resistance of BE63/3. Bioinformatics showed 125 de-regulated genes that, according to enrichment analysis, were implicated in collagen synthesis/metabolism. Connective tissue growth factor (CTGF) was one of the higher up-regulated mRNA. Five nM EVE was ineffective on the pro-fibrotic machinery. Additionally, 3 miRNAs resulted hyper-expressed after 100 nM EVE and able to regulate 31 of the genes selected by the transcriptomic analysis (including CTGF). RT-PCR and western blot for MMP12 and CTGF validated high-throughput results. Our results revealed a complex biological network implicated in EVE-related pulmonary fibrosis and underlined new potential disease biomarkers and therapeutic targets.
Keywords: epithelial to mesenchymal transition; everolimus; mTOR inhibitor; miRNome; pulmonary fibrosis; transcriptomics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

