Tracking disease progression non-invasively in Duchenne and Becker muscular dystrophies
- PMID: 29682908
- PMCID: PMC6104105
- DOI: 10.1002/jcsm.12304
Tracking disease progression non-invasively in Duchenne and Becker muscular dystrophies
Abstract
Background: Analysis of muscle biopsies allowed to characterize the pathophysiological changes of Duchenne and Becker muscular dystrophies (D/BMD) leading to the clinical phenotype. Muscle tissue is often investigated during interventional dose finding studies to show in situ proof of concept and pharmacodynamics effect of the tested drug. Less invasive readouts are needed to objectively monitor patients' health status, muscle quality, and response to treatment. The identification of serum biomarkers correlating with clinical function and able to anticipate functional scales is particularly needed for personalized patient management and to support drug development programs.
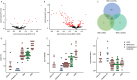
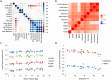
Methods: A large-scale proteomic approach was used to identify serum biomarkers describing pathophysiological changes (e.g. loss of muscle mass), association with clinical function, prediction of disease milestones, association with in vivo 31 P magnetic resonance spectroscopy data and dystrophin levels in muscles. Cross-sectional comparisons were performed to compare DMD patients, BMD patients, and healthy controls. A group of DMD patients was followed up for a median of 4.4 years to allow monitoring of individual disease trajectories based on yearly visits.
Results: Cross-sectional comparison enabled to identify 10 proteins discriminating between healthy controls, DMD and BMD patients. Several proteins (285) were able to separate DMD from healthy, while 121 proteins differentiated between BMD and DMD; only 13 proteins separated BMD and healthy individuals. The concentration of specific proteins in serum was significantly associated with patients' performance (e.g. BMP6 serum levels and elbow flexion) or dystrophin levels (e.g. TIMP2) in BMD patients. Analysis of longitudinal trajectories allowed to identify 427 proteins affected over time indicating loss of muscle mass, replacement of muscle by adipose tissue, and cardiac involvement. Over-representation analysis of longitudinal data allowed to highlight proteins that could be used as pharmacodynamic biomarkers for drugs currently in clinical development.
Conclusions: Serum proteomic analysis allowed to not only discriminate among DMD, BMD, and healthy subjects, but it enabled to detect significant associations with clinical function, dystrophin levels, and disease progression.
Keywords: Becker muscular dystrophy; Biomarker; Duchenne muscular dystrophy; Longitudinal analysis; Proteomics; Sarcopenia.
© 2018 The Authors. Journal of Cachexia, Sarcopenia and Muscle published by John Wiley & Sons Ltd on behalf of the Society on Sarcopenia, Cachexia and Wasting Disorders.
Figures


References
-
- Manzur AY, Muntoni F. Diagnosis and new treatments in muscular dystrophies. J Neurol Neurosurg Psychiatry 2009;80:706–714. - PubMed
-
- Ellis JA, Vroom E, Muntoni F. 195th ENMC International Workshop: newborn screening for Duchenne muscular dystrophy 14–16th December, 2012, Naarden, The Netherlands. Neuromuscul Disord 2013;23:682–689. - PubMed
-
- Way M, Pope B, Cross RA, Kendrick‐Jones J, Weeds AG. Expression of the N‐terminal domain of dystrophin in E. coli and demonstration of binding to F‐actin. FEBS Lett 1992;301:243–245. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

