TCRβ Combinatorial Immunoreceptor Expression by Neutrophils Correlates with Parasite Burden and Enhanced Phagocytosis during a Plasmodium berghei ANKA Malaria Infection
- PMID: 29685989
- PMCID: PMC6013678
- DOI: 10.1128/IAI.00899-17
TCRβ Combinatorial Immunoreceptor Expression by Neutrophils Correlates with Parasite Burden and Enhanced Phagocytosis during a Plasmodium berghei ANKA Malaria Infection
Abstract
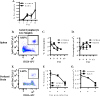
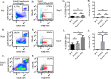
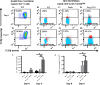
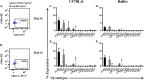
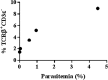
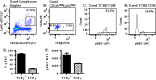
Recent studies have demonstrated that a subpopulation of neutrophils express the TCRαβ combinatorial immunoreceptor in humans and mice. Here, we report that a Plasmodium berghei ANKA murine malaria infection induces expansion of TCRβ expressing CD11b+ Ly6G+ neutrophils in the spleen during the early phase of infection. Measurement of TCRβ transcript and protein levels of neutrophils in wild-type versus nude and Rag1 knockout mice establishes that the observed expression is not a consequence of nonspecific antibody staining or passive receptor expression due to phagocytosis or trogocytosis of peripheral T cells. Remarkably, on day 3 postinfection, we observed a highly significant correlation between the proportion of neutrophils that express TCRβ and peripheral blood parasite burden. In addition, TCRβ+ neutrophils phagocytose parasitized erythrocytes with 4-fold greater efficiency than TCRβ- neutrophils. Together these results signify that TCR expression by the neutrophil plays an important role in the regulation of parasite burden by enhancing the phagocytic capacity of the neutrophil.
Keywords: Plasmodium berghei ANKA; TCRβ; neutrophil.
This is a work of the U.S. Government and is not subject to copyright protection in the United States. Foreign copyrights may apply.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

