Prevention of hepatocellular carcinoma by targeting MYCN-positive liver cancer stem cells with acyclic retinoid
- PMID: 29686061
- PMCID: PMC5949003
- DOI: 10.1073/pnas.1802279115
Prevention of hepatocellular carcinoma by targeting MYCN-positive liver cancer stem cells with acyclic retinoid
Abstract
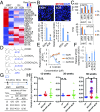
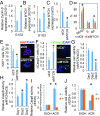
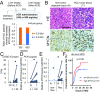
Hepatocellular carcinoma (HCC) is a highly lethal cancer that has a high rate of recurrence, in part because of cancer stem cell (CSC)-dependent field cancerization. Acyclic retinoid (ACR) is a synthetic vitamin A-like compound capable of preventing the recurrence of HCC. Here, we performed a genome-wide transcriptome screen and showed that ACR selectively suppressed the expression of MYCN, a member of the MYC family of basic helix-loop-helix-zipper transcription factors, in HCC cell cultures, animal models, and liver biopsies obtained from HCC patients. MYCN expression in human HCC was correlated positively with both CSC and Wnt/β-catenin signaling markers but negatively with mature hepatocyte markers. Functional analysis showed repressed cell-cycle progression, proliferation, and colony formation, activated caspase-8, and induced cell death in HCC cells following silencing of MYCN expression. High-content single-cell imaging analysis and flow cytometric analysis identified a MYCN+ CSC subpopulation in the heterogeneous HCC cell cultures and showed that these cells were selectively killed by ACR. Particularly, EpCAM+ cells isolated using a cell-sorting system showed increased MYCN expression and sensitivity to ACR compared with EpCAM- cells. In a long-term (>10 y) follow-up study of 102 patients with HCC, MYCN was expressed at higher levels in the HCC tumor region than in nontumor regions, and there was a positive correlation between MYCN expression and recurrence of de novo HCC but not metastatic HCC after curative treatment. In summary, these results suggest that MYCN serves as a prognostic biomarker and therapeutic target of ACR for liver CSCs in de novo HCC.
Keywords: MYCN; acyclic retinoid; cancer stem cell; hepatocellular carcinoma; transcriptome.
Copyright © 2018 the Author(s). Published by PNAS.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Comment in
-
Reply to Yoshida: Liver cancer stem cells: Identification and lipid metabolic reprogramming.Proc Natl Acad Sci U S A. 2018 Jul 10;115(28):E6390-E6391. doi: 10.1073/pnas.1808740115. Epub 2018 Jun 25. Proc Natl Acad Sci U S A. 2018. PMID: 29941607 Free PMC article. No abstract available.
-
How to eliminate MYCN-positive hepatic cancer stem cells to prevent the recurrence?Proc Natl Acad Sci U S A. 2018 Jul 10;115(28):E6388-E6389. doi: 10.1073/pnas.1808092115. Epub 2018 Jun 25. Proc Natl Acad Sci U S A. 2018. PMID: 29941608 Free PMC article. No abstract available.
References
-
- Venook AP, Papandreou C, Furuse J, de Guevara LL. The incidence and epidemiology of hepatocellular carcinoma: A global and regional perspective. Oncologist. 2010;15:5–13. - PubMed
-
- Perz JF, Armstrong GL, Farrington LA, Hutin YJ, Bell BP. The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J Hepatol. 2006;45:529–538. - PubMed
-
- Yoshimoto S, et al. Obesity-induced gut microbial metabolite promotes liver cancer through senescence secretome. Nature. 2013;499:97–101, and erratum (2014) 506:396. - PubMed
-
- Kang TW, et al. Senescence surveillance of pre-malignant hepatocytes limits liver cancer development. Nature. 2011;479:547–551. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

