Population Pharmacokinetic Model for Vancomycin Used in Open Heart Surgery: Model-Based Evaluation of Standard Dosing Regimens
- PMID: 29686154
- PMCID: PMC6021682
- DOI: 10.1128/AAC.00088-18
Population Pharmacokinetic Model for Vancomycin Used in Open Heart Surgery: Model-Based Evaluation of Standard Dosing Regimens
Abstract
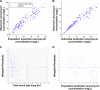
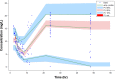
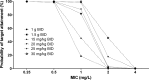
The purpose of this study was to investigate the population pharmacokinetics of vancomycin in patients undergoing open heart surgery. In this observational pharmacokinetic study, multiple blood samples were drawn over a 48-h period of intravenous vancomycin in patients who were undergoing open heart surgery. Blood samples were analyzed using an Architect i4000SR immunoassay analyzer. Population pharmacokinetic models were developed using Monolix 4.4 software. Pharmacokinetic-pharmacodynamic (PK-PD) simulations were performed to explore the ability of different dosage regimens to achieve the pharmacodynamic targets. A total of 168 blood samples were analyzed from 28 patients. The pharmacokinetics of vancomycin are best described by a two-compartment model with between-subject variability in clearance (CL), the volume of distribution of the central compartment (V1), and volume of distribution of the peripheral compartment (V2). The CL and the V1 of vancomycin were related to creatinine CL (CLCR), body weight, and albumin concentration. Dosing simulations showed that standard dosing regimens of 1 and 1.5 g failed to achieve the PK-PD target of AUC0-24/MIC > 400 for an MIC of 1 mg/liter, while high weight-based dosing regimens were able to achieve the PK-PD target. In summary, the administration of standard doses of 1 and 1.5 g of vancomycin two times daily provided inadequate antibiotic prophylaxis in patients undergoing open heart surgery. The same findings were obtained when 15- and 20-mg/kg doses of vancomycin were administered. Achieving the PK-PD target required higher doses (25 and 30 mg/kg) of vancomycin.
Keywords: Monte Carlo simulation; PK-PD; open heart surgery; population pharmacokinetics; prophylaxis; vancomycin.
Copyright © 2018 American Society for Microbiology.
Figures
References
-
- Branch-Elliman W, Ripollone JE, O'Brien WJ, Itani KMF, Schweizer ML, Perencevich E, Strymish J, Gupta K. 2017. Risk of surgical site infection, acute kidney injury, and Clostridium difficile infection following antibiotic prophylaxis with vancomycin plus a beta-lactam versus either drug alone: a national propensity-score-adjusted retrospective cohort study. PLoS Med 14:e1002340. doi: 10.1371/journal.pmed.1002340. - DOI - PMC - PubMed
-
- Reineke S, Carrel TP, Eigenmann V, Gahl B, Fuehrer U, Seidl C, Reineke D, Roost E, Bachli M, Marschall J, Englberger L. 2017. Adding vancomycin to perioperative prophylaxis decreases deep sternal wound infections in high-risk cardiac surgery patients. Eur J Cardiothorac Surg doi: 10.1093/ejcts/ezx328. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

