Estradiol alters body temperature regulation in the female mouse
- PMID: 29687044
- PMCID: PMC5902204
- DOI: 10.1080/23328940.2017.1384090
Estradiol alters body temperature regulation in the female mouse
Abstract
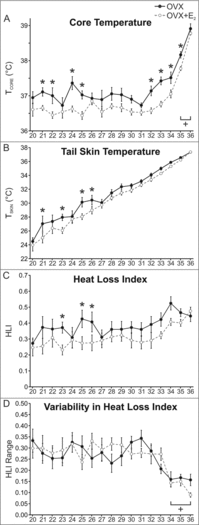
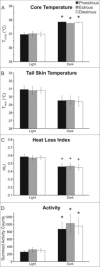
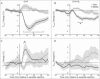
Hot flushes are due to estrogen withdrawal and characterized by the episodic activation of heat dissipation effectors. Recent studies (in humans and rats) have implicated neurokinin 3 (NK3) receptor signaling in the genesis of hot flushes. Although transgenic mice are increasingly used for biomedical research, there is limited information on how 17β-estradiol and NK3 receptor signaling alters thermoregulation in the mouse. In this study, a method was developed to measure tail skin temperature (TSKIN) using a small data-logger attached to the surface of the tail, which, when combined with a telemetry probe for core temperature (TCORE), allowed us to monitor thermoregulation in freely-moving mice over long durations. We report that estradiol treatment of ovariectomized mice reduced TCORE during the light phase (but not the dark phase) while having no effect on TSKIN or activity. Estradiol also lowered TCORE in mice exposed to ambient temperatures ranging from 20 to 36°C. Unlike previous studies in the rat, estradiol treatment of ovariectomized mice did not reduce TSKIN during the dark phase. Subcutaneous injections of an NK3 receptor agonist (senktide) in ovariectomized mice caused an acute increase in TSKIN and a reduction in TCORE, consistent with the activation of heat dissipation effectors. These changes were reduced by estradiol, suggesting that estradiol lowers the sensitivity of central thermoregulatory pathways to NK3 receptor activation. Overall, we show that estradiol treatment of ovariectomized mice decreases TCORE during the light phase, reduces the thermoregulatory effects of senktide and modulates thermoregulation differently than previously described in the rat.
Keywords: estrogen; hot flush; kisspeptin; menopause; neurokinin B; vasodilation.
Figures
Similar articles
-
Neurokinin 3 Receptor-Expressing Neurons in the Median Preoptic Nucleus Modulate Heat-Dissipation Effectors in the Female Rat.Endocrinology. 2015 Jul;156(7):2552-62. doi: 10.1210/en.2014-1974. Epub 2015 Mar 31. Endocrinology. 2015. PMID: 25825817 Free PMC article.
-
Effects of estradiol on the thermoneutral zone and core temperature in ovariectomized rats.Endocrinology. 2010 Mar;151(3):1187-93. doi: 10.1210/en.2009-1112. Epub 2010 Jan 5. Endocrinology. 2010. PMID: 20051485 Free PMC article.
-
Activation of neurokinin 3 receptors in the median preoptic nucleus decreases core temperature in the rat.Endocrinology. 2011 Dec;152(12):4894-905. doi: 10.1210/en.2011-1492. Epub 2011 Oct 25. Endocrinology. 2011. PMID: 22028440 Free PMC article.
-
The neuroendocrinology of the preoptic area in menopause: Symptoms and therapeutic strategies.Handb Clin Neurol. 2021;179:455-460. doi: 10.1016/B978-0-12-819975-6.00029-7. Handb Clin Neurol. 2021. PMID: 34225982 Review.
-
Ergogenic effects of precooling with cold water immersion and ice ingestion: A meta-analysis.Eur J Sport Sci. 2018 Mar;18(2):170-181. doi: 10.1080/17461391.2017.1405077. Epub 2017 Nov 26. Eur J Sport Sci. 2018. PMID: 29173092 Review.
Cited by
-
Invited review: Translating kisspeptin and neurokinin B biology into new therapies for reproductive health.J Neuroendocrinol. 2022 Oct;34(10):e13201. doi: 10.1111/jne.13201. Epub 2022 Oct 19. J Neuroendocrinol. 2022. PMID: 36262016 Free PMC article. Review.
-
The Effects of Estrogens on Neural Circuits That Control Temperature.Endocrinology. 2021 Aug 1;162(8):bqab087. doi: 10.1210/endocr/bqab087. Endocrinology. 2021. PMID: 33939822 Free PMC article. Review.
-
Deletion of Growth Hormone Secretagogue Receptor in Kisspeptin Neurons in Female Mice Blocks Diet-Induced Obesity.Biomolecules. 2022 Sep 25;12(10):1370. doi: 10.3390/biom12101370. Biomolecules. 2022. PMID: 36291579 Free PMC article.
-
Influences of ovarian hormones on physiological responses to cold in women.Temperature (Austin). 2021 Sep 14;9(1):23-45. doi: 10.1080/23328940.2021.1953688. eCollection 2022. Temperature (Austin). 2021. PMID: 35655670 Free PMC article. Review.
-
Estrogen-sensitive medial preoptic area neurons coordinate torpor in mice.Nat Commun. 2020 Dec 11;11(1):6378. doi: 10.1038/s41467-020-20050-1. Nat Commun. 2020. PMID: 33311503 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
