The Taxonomy of Covalent Inhibitors
- PMID: 29689165
- PMCID: PMC6016374
- DOI: 10.1021/acs.biochem.8b00315
The Taxonomy of Covalent Inhibitors
Abstract
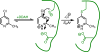
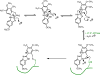
Covalent enzyme inhibitors are widely applied as biochemical tools and therapeutic agents. As a complement to categorization of these inhibitors by reactive group or modification site, we present a categorization by mechanism, which highlights common advantages and disadvantages inherent to each approach. Established categories for reversible and irreversible covalent inhibition are reviewed with representative examples given for each class, including covalent reversible inhibitors, slow substrates, residue-specific reagents, affinity labels (classical, quiescent, and photoaffinity), and mechanism-based inactivators. The relationships of these categories to proteomic profiling probes (activity-based and reactivity-based) as well as complementary approaches such as prodrug and soft drug design are also discussed. A wide variety of strategies are used to balance reactivity and selectivity in the design of covalent enzyme inhibitors. Use of a shared terminology is encouraged to clearly convey these mechanisms, to relate them to prior use of covalent inhibitors in enzymology, and to facilitate the development of more effective covalent inhibitors.
Conflict of interest statement
The authors declare no competing financial Interest.
Figures

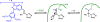


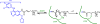

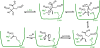



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

