Time and temperature dependent analytical stability of dry-collected Evalyn HPV self-sampling brush for cervical cancer screening
- PMID: 29689311
- PMCID: PMC6026099
- DOI: 10.1016/j.pvr.2018.04.005
Time and temperature dependent analytical stability of dry-collected Evalyn HPV self-sampling brush for cervical cancer screening
Abstract
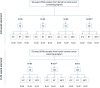
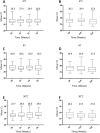
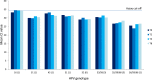
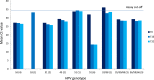
As a new initiative, HPV self-sampling to non-attenders using the dry Evalyn self-sampling brush is offered in the Capital Region of Denmark. The use of a dry brush is largely uncharted territory in terms of analytical stability. In this study we aim to provide evidence on the analytical quality of dry HPV self-sampling brushes as a function of time and temperature. We assessed the analytical stability of dry stored Evalyn brushes at three different temperatures, (4 °C, room temperature, 30 °C) and five different storage time points; T = 0 (baseline), 2, 4, 8, 16, and 32 weeks prior to HPV analysis using the BD Onclarity HPV assay. Mean Ct value of the Onclarity internal control was used as comparator of cellularity across time and temperatures, with no or only borderline statistical differences observed. HPV detection was stable throughout the five time points. In addition, analytically amplifiable DNA copy numbers and DNA fragmentation was assessed using the Agena iPLEX Exome QC assay, with no or only borderline statistical differences observed. In conclusion, the Evalyn brush is analytically stable with respect to human genomic material and HPV detection for up to 32 weeks at temperatures ranging from 4 °C to 30 °C.
Keywords: Analytical stability; Cervical cancer screening; Dry self-sampling brushes; HPV; Self-sampling.
Copyright © 2018 The Authors. Published by Elsevier B.V. All rights reserved.
Figures
Similar articles
-
Direct comparison of two vaginal self-sampling devices for the detection of human papillomavirus infections.J Clin Virol. 2016 Sep;82:46-50. doi: 10.1016/j.jcv.2016.06.016. Epub 2016 Jun 28. J Clin Virol. 2016. PMID: 27434147
-
Excellent analytical and clinical performance of a dry self-sampling device for human papillomavirus detection in an urban Chinese referral population.J Obstet Gynaecol Res. 2016 Dec;42(12):1839-1845. doi: 10.1111/jog.13132. Epub 2016 Sep 20. J Obstet Gynaecol Res. 2016. PMID: 27647799
-
Validation of BD Onclarity HPV Assay on Vaginal Self-Samples versus Cervical Samples Using the VALHUDES Protocol.Cancer Epidemiol Biomarkers Prev. 2022 Dec 5;31(12):2177-2184. doi: 10.1158/1055-9965.EPI-22-0757. Cancer Epidemiol Biomarkers Prev. 2022. PMID: 36099441
-
The potential role of self-sampling for high-risk human papillomavirus detection in cervical cancer screening.Rev Med Virol. 2011 May;21(3):139-53. doi: 10.1002/rmv.686. Rev Med Virol. 2011. PMID: 21538664 Review.
-
Emerging role of HPV self-sampling in cervical cancer screening for hard-to-reach women: Focused literature review.Can Fam Physician. 2017 Aug;63(8):597-601. Can Fam Physician. 2017. PMID: 28807952 Free PMC article. Review.
Cited by
-
An observational study comparing HPV prevalence and type distribution between HPV-vaccinated and -unvaccinated girls after introduction of school-based HPV vaccination in Norway.PLoS One. 2019 Oct 10;14(10):e0223612. doi: 10.1371/journal.pone.0223612. eCollection 2019. PLoS One. 2019. PMID: 31600341 Free PMC article.
-
Evaluation of the concordance in HPV type between self- and physician-collected samples using a brush-based device and a PCR-based HPV DNA test in Japanese referred patients with abnormal cytology or HPV infection.Int J Clin Oncol. 2020 Oct;25(10):1854-1860. doi: 10.1007/s10147-020-01727-5. Epub 2020 Jun 24. Int J Clin Oncol. 2020. PMID: 32583223 Free PMC article.
-
Design of a pragmatic randomized controlled trial of home-based human papillomavirus (HPV) self-sampling for increasing cervical cancer screening uptake in a U.S. healthcare system: The STEP trial.Contemp Clin Trials. 2022 Nov;122:106960. doi: 10.1016/j.cct.2022.106960. Epub 2022 Oct 12. Contemp Clin Trials. 2022. PMID: 36241145 Free PMC article. Clinical Trial.
-
Evaluating Opt-In Vaginal Human Papillomavirus Self-Sampling: Participation Rates and Detection of High-Grade Lesions (CIN2+) among Unscreened Japanese Women Aged 30-39.Healthcare (Basel). 2024 Mar 6;12(5):599. doi: 10.3390/healthcare12050599. Healthcare (Basel). 2024. PMID: 38470710 Free PMC article.
-
Genital self-sampling for HPV-based cervical cancer screening: a qualitative study of preferences and barriers in rural Ethiopia.BMC Public Health. 2019 Jul 31;19(1):1026. doi: 10.1186/s12889-019-7354-4. BMC Public Health. 2019. PMID: 31366402 Free PMC article.
References
-
- Bulkmans N.W., Berkhof J., Rozendaal L., van Kemenade F.J., Boeke A.J., Bulk S., Voorhorst F.J., Verheijen R.H., van Groningen K., Boon M.E. Human papillomavirus DNA testing for the detection of cervical intraepithelial neoplasia grade 3 and cancer: 5-year follow-up of a randomised controlled implementation trial. Lancet. 2007;370(9601):1764–1772. - PubMed
-
- Kitchener H.C., Almonte M., Gilham C., Dowie R., Stoykova B., Sargent A., Roberts C., Desai M., Peto J., Group A.T.S. ARTISTIC: a randomised trial of human papillomavirus (HPV) testing in primary cervical screening. Health Technol. Assess. 2009;13(51):1–150. (iii-iv) - PubMed
-
- Leinonen M., Nieminen P., Kotaniemi-Talonen L., Malila N., Tarkkanen J., Laurila P., Anttila A. Age-specific evaluation of primary human papillomavirus screening vs conventional cytology in a randomized setting. J. Natl. Cancer Inst. 2009;101(23):1612–1623. - PubMed
-
- Mayrand M.H., Duarte-Franco E., Rodrigues I., Walter S.D., Hanley J., Ferenczy A., Ratnam S., Coutlee F., Franco E.L. Canadian Cervical Cancer Screening Trial Study G: human papillomavirus DNA versus Papanicolaou screening tests for cervical cancer. N. Engl. J. Med. 2007;357(16):1579–1588. - PubMed
-
- Naucler P., Ryd W., Tornberg S., Strand A., Wadell G., Elfgren K., Radberg T., Strander B., Johansson B., Forslund O. Human papillomavirus and Papanicolaou tests to screen for cervical cancer. N. Engl. J. Med. 2007;357(16):1589–1597. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

