Carnitine Acetyltransferase in AgRP Neurons Is Required for the Homeostatic Adaptation to Restricted Feeding in Male Mice
- PMID: 29697769
- PMCID: PMC6692886
- DOI: 10.1210/en.2018-00131
Carnitine Acetyltransferase in AgRP Neurons Is Required for the Homeostatic Adaptation to Restricted Feeding in Male Mice
Abstract
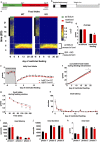
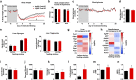
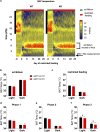
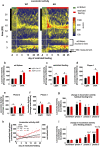
Behavioral adaptation to periods of varying food availability is crucial for survival, and agouti-related protein (AgRP) neurons have been associated with entrainment to temporal restricted feeding. We have shown that carnitine acetyltransferase (Crat) in AgRP neurons enables metabolic flexibility and appropriate nutrient partitioning. In this study, by restricting food availability to 3 h/d during the light phase, we examined whether Crat is a component of a food-entrainable oscillator (FEO) that helps link behavior to food availability. AgRP Crat knockout (KO) mice consumed less food and regained less body weight but maintained blood glucose levels during the 25-day restricted feeding protocol. Importantly, we observed no difference in meal latency, food anticipatory activity (FAA), or brown adipose tissue temperature during the first 13 days of restricted feeding. However, as the restricted feeding paradigm progressed, we noticed an increased FAA in AgRP Crat KO mice. The delayed increase in FAA, which developed during the last 12 days of restricted feeding, corresponded with elevated plasma levels of corticosterone and nonesterified fatty acids, indicating it resulted from greater energy debt incurred by KO mice over the course of the experiment. These experiments highlight the importance of Crat in AgRP neurons in regulating feeding behavior and body weight gain during restricted feeding but not in synchronizing behavior to food availability. Thus, Crat within AgRP neurons forms a component of the homeostatic response to restricted feeding but is not likely to be a molecular component of FEO.
Figures
References
-
- Carneiro BT, Araujo JF. The food-entrainable oscillator: a network of interconnected brain structures entrained by humoral signals? Chronobiol Int. 2009;26(7):1273–1289. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

