Chromosomal integrity and DNA damage in freeze-dried spermatozoa
- PMID: 29699095
- PMCID: PMC5907138
- DOI: 10.1007/s12522-011-0092-7
Chromosomal integrity and DNA damage in freeze-dried spermatozoa
Abstract
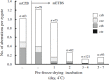
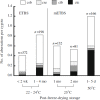
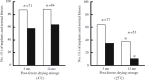
Freeze-drying technology may one day be used to preserve mammalian spermatozoa indefinitely without cryopreservation. Freeze-dried mouse spermatozoa stored below 4°C for up to 1 year have maintained the ability to fertilize oocytes and support normal development. The maximum storage period for spermatozoa increases at lower storage temperatures. Freeze-drying, per se, may reduce the integrity of chromosomes in freeze-dried mouse spermatozoa, but induction of chromosomal damage is suppressed if spermatozoa are incubated with divalent cation chelating agents prior to freeze-drying. Nevertheless, chromosomal damage does accumulate in spermatozoa stored at temperatures above 4°C. Currently, no established methods or strategies can prevent or reduce damage accumulation, and damage accumulation during storage is a serious obstacle to advances in freeze-drying technology. Chromosomal integrity of freeze-dried human spermatozoa have roughly background levels of chromosomal damage after storage at 4°C for 1 month, but whether these spermatozoa can produce healthy newborns is unknown. The safety of using freeze-dried human spermatozoa must be evaluated based on the risks of heritable chromosome and DNA damage that accumulates during storage.
Keywords: Chromosome; Cryopreservation; DNA damage; Freeze‐drying; Spermatozoa.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
