Synthesis and conformational analysis of linear homo- and heterooligomers from novel 2-C-branched sugar amino acids (SAAs)
- PMID: 29700416
- PMCID: PMC5919921
- DOI: 10.1038/s41598-018-24927-6
Synthesis and conformational analysis of linear homo- and heterooligomers from novel 2-C-branched sugar amino acids (SAAs)
Abstract
Sugar amino acids (SAAs), as biologically interesting structures bearing both amino and carboxylic acid functional groups represent an important class of multifunctional building blocks. In this study, we develop an easy access to novel SAAs in only three steps starting from nitro compounds in high yields in analytically pure form, easily available by ceric (IV) mediated radical additions. Such novel SAAs have been applied in the assembly of total nine carbopeptoids with the form of linear homo- and heterooligomers for the structural investigations employing circular dichroism (CD) spectroscopy, which suggest that the carbopeptoids emerge a well-extended, left (or right)-handed conformation similar to polyproline II (PPII) helices. NMR studies also clearly demonstrated the presence of ordered secondary structural elements. 2D-ROESY spectra were acquired to identify i+1 NH ↔ i C 1 H, i C 2 H correlations which support the conformational analysis of tetramers by CD spectroscopy. These findings provide interesting information of SAAs and their oligomers as potential scaffolds for discovering new drugs and materials.
Conflict of interest statement
The authors declare no competing interests.
Figures

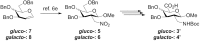





References
-
- Gervay-Hague J, Weathers TM. Pyranosyl Sugar Amino Acids Conjugates: Their BiologicalOrigins, Synthetic Preparations, and Structural Characterization. J. Carbohydr. Chem. 2002;21:867–910. doi: 10.1081/CAR-120016491. - DOI
-
- Chakraborty TK, Jayaprakash S, Ghosh S. Sugar Amino Acid Based Scaffolds - Novel Peptidomimetics and their Potential in CombinatorialSynthesis. Comb. Chem. High. T. Scr. 2002;5:373–387. - PubMed
-
- Schweizer F. Unusual Amino Acids Accessed Through Sugar-Amino Acid Hybrids and Incorporation into Biologically Active Peptides. Trends Glycosci. Glycotechnol. 2003;15:315–328. doi: 10.4052/tigg.15.315. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

