Oxygen-dependence of mitochondrial ROS production as detected by Amplex Red assay
- PMID: 29702406
- PMCID: PMC6007170
- DOI: 10.1016/j.redox.2018.04.014
Oxygen-dependence of mitochondrial ROS production as detected by Amplex Red assay
Abstract
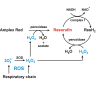
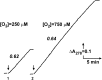
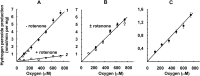
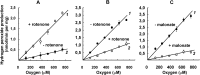
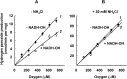
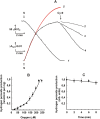
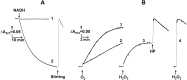
The initial rates of superoxide plus hydrogen peroxide (ROS) generation by intact or permeabilized rat heart mitochondria and coupled inside-out bovine heart submitochondrial particles (SMP) oxidizing NAD-dependent substrates, NADH, and succinate were measured by detecting resorufin formation in the Amplex Red assay at various oxygen concentrations. Linear dependences of the initial rates on oxygen concentration within the range of ~125-750 μM were found for all significant mitochondrial generators, i.e. the respiratory complexes and ammonium-stimulated dihydrolipoamide dehydrogenase. At lower oxygen concentrations upon its decrease from air saturation level to zero, the time-course of resorufin formation by SMP catalyzing coupled oxidation of succinate (the total ROS production by respiratory complexes II and III and by the reverse electron transfer (RET)-mediated by complex I) also corresponds to the linear dependence on oxygen with the same first-order rate constant determined in the initial rate studies. Prolonged incubation of SMP generating succinate-supported complex I-mediated ROS affected neither their NADH oxidase nor ROS generating activity. In contrast to SMP significant deviation from the first-order oxygen dependence in the time-course kinetics during coupled oxidation of succinate by intact mitochondria was evident. Complex I catalyzes the NADH:resorufin oxidoreductase reaction resulting in formation of colorless reduced resorufin. Hydrogen peroxide oxidizes reduced resorufin in the presence of peroxidase, thus showing its dihydroresorufin peroxidase activity. Combined NADH:resorufin reductase and dihydroresorufin peroxidase activities result in underestimation of the amount of hydrogen peroxide generated by mitochondria. We conclude that only initial rates of the mitochondrial ROS production, not the amount of resorufin accumulated, should be taken as the reliable measure of the mitochondrial ROS-generating activity, because of the cycling of the oxidized and reduced resorufin during Amplex Red assays fed by NADH and other possible reductant(s) present in mitochondria.
Keywords: Amplex Red; Hydrogen peroxide; Mitochondria; Resorufin; Respiratory chain; Respiratory complex I.
Copyright © 2018 The Authors. Published by Elsevier B.V. All rights reserved.
Figures

References
-
- Gerschman R., Gilbert D.L., Nye S.W., Dwyer P., Fenn W.O. Oxygen poisoning and X-irradiation: a mechanism in common. Science. 1954;119(3097):623–626. - PubMed
-
- Jensen P.K. Antimycin-insensitive oxidation of succinate and reduced nicotinamide-adenine dinucleotide in electron-transport particles. Biochim. Biophys. Acta. 1966;122(2):157–166. - PubMed
-
- Chance B., Sies H., Boveris A. Hydroperoxide metabolism in mammalian organs. Physiol. Rev. 1979;59:527–605. - PubMed
-
- Cadenas E., Davies K.J. Mitochondrial free radical generation, oxidative stress, and aging. Free Radic. Biol. Med. 2000;29(3–4):222–230. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

