Theranostics and metabolotheranostics for precision medicine in oncology
- PMID: 29705040
- PMCID: PMC5943142
- DOI: 10.1016/j.jmr.2018.03.004
Theranostics and metabolotheranostics for precision medicine in oncology
Abstract
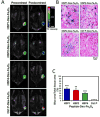
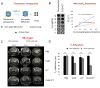
Most diseases, especially cancer, would significantly benefit from precision medicine where treatment is shaped for the individual. The concept of theragnostics or theranostics emerged around 2002 to describe the incorporation of diagnostic assays into the selection of therapy for this purpose. Increasingly, theranostics has been used for strategies that combine noninvasive imaging-based diagnostics with therapy. Within the past decade theranostic imaging has transformed into a rapidly expanding field that is located at the interface of diagnosis and therapy. A critical need in cancer treatment is to minimize damage to normal tissue. Molecular imaging can be applied to identify targets specific to cancer with imaging, design agents against these targets to visualize their delivery, and monitor response to treatment, with the overall purpose of minimizing collateral damage. Genomic and proteomic profiling can provide an extensive 'fingerprint' of each tumor. With this cancer fingerprint, theranostic agents can be designed to personalize treatment for precision medicine of cancer, and minimize damage to normal tissue. Here, for the first time, we have introduced the term 'metabolotheranostics' to describe strategies where disease-based alterations in metabolic pathways detected by MRS are specifically targeted with image-guided delivery platforms to achieve disease-specific therapy. The versatility of MRI and MRS in molecular and functional imaging makes these technologies especially important in theranostic MRI and 'metabolotheranostics'. Our purpose here is to provide insights into the capabilities and applications of this exciting new field in cancer treatment with a focus on MRI and MRS.
Keywords: Cancer; Metabolotheranostics; Precision medicine; Theranostics.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures







References
-
- Rivlin M, Navon G. CEST MRI of 3-O-methyl-D-glucose on different breast cancer models. Magn Reson Med. 2017 - PubMed
-
- Xu X, Yadav NN, Knutsson L, Hua J, Kalyani R, Hall E, Laterra J, Blakeley J, Strowd R, Pomper M, Barker P, Chan K, Liu G, McMahon MT, Stevens RD, van Zijl PC. Dynamic Glucose-Enhanced (DGE) MRI: Translation to Human Scanning and First Results in Glioma Patients. Tomography. 2015;1(2):105–14. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

