Intestinal absorption of glucose in mice as determined by positron emission tomography
- PMID: 29707805
- PMCID: PMC6023830
- DOI: 10.1113/JP275934
Intestinal absorption of glucose in mice as determined by positron emission tomography
Abstract
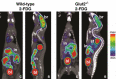
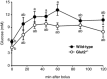
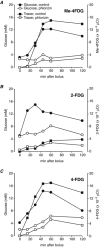
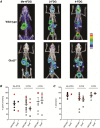
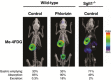
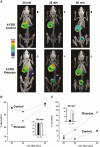
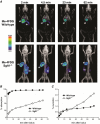
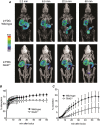
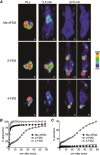
Key points: The goal was to determine the importance of the sodium-glucose cotransporter SGLT1 and the glucose uniporter GLUT2 in intestinal glucose absorption during oral glucose tolerance tests (OGTTs) in mice. Glucose absorption was determined in mice using positron emission tomography and three non-metabolizable glucose probes: one specific for SGLTs, one specific for GLUTs, and one a substrate for both SGLTs and GLUTs. Absorption was determined in wild-type, Sglt1-/- and Glut2-/- mice. Gastric emptying was a rate-limiting step in absorption. SGLT1, but not GLUT2, was important in fast glucose absorption. In the absence of SGLT1 or GLUT2, the oral glucose load delivered to the small intestine was slowly absorbed. Oral phlorizin only inhibited the fast component of glucose absorption, but it contributed to decreasing blood glucose levels by inhibiting renal reabsorption.
Abstract: The current model of intestinal absorption is that SGLT1 is responsible for transport of glucose from the lumen into enterocytes across the brush border membrane, and GLUT2 for the downhill transport from the epithelium into blood across the basolateral membrane. Nevertheless, questions remain about the importance of these transporters in vivo. To address these questions, we have developed a non-invasive imaging method, positron emission tomography (PET), to monitor intestinal absorption of three non-metabolized glucose tracers during standard oral glucose tolerance tests (OGTTs) in mice. One tracer is specific for SGLTs (α-methyl-4-[18 F]fluoro-4-deoxy-d-glucopyranoside; Me-4FDG), one is specific for GLUTs (2-deoxy-2-[18 F]fluoro-d-glucose; 2-FDG), and one is a substrate for both SGLTs and GLUTs (4-deoxy-4-[18 F]fluoro-d-glucose; 4-FDG). OGTTs were conducted on adult wild-type, Sglt1-/- and Glut2-/- mice. In conscious mice, OGTTs resulted in the predictable increase in blood glucose that was blocked by phlorizin in both wild-type and Glut2-/- animals. The blood activity of both Me-4FDG and 4-FDG, but not 2-FDG, accompanied the changes in glucose concentration. PET imaging during OGTTs further shows that: (i) intestinal absorption of the glucose load depends on gastric emptying; (ii) SGLT1 is important for the fast absorption; (iii) GLUT2 is not important in absorption; and (iv) oral phlorizin reduces absorption by SGLT1, but is absorbed and blocks glucose reabsorption in the kidney. We conclude that in standard OGTTs in mice, SGLT1 is essential in fast absorption, GLUT2 does not play a significant role, and in the absence of SGLT1 the total load of glucose is slowly absorbed.
Keywords: PET; absorption; intestine.
© 2018 The Authors. The Journal of Physiology published by John Wiley & Sons Ltd on behalf of The Physiological Society.
Figures
References
-
- Bindslev N & Wright EM (1976). Effect of temperature on nonelectrolyte permeation across the toad urinary bladder. J Membr Biol 29, 265–288. - PubMed
-
- Casteleyn C, Rekecki A, Van der Aa A, Simoens P & Van den Broeck W (2010). Surface area assessment of the murine intestinal tract as a prerequisite for oral dose translation from mouse to man. Lab Anim 44, 176–183. - PubMed

