Memory Allocation: Mechanisms and Function
- PMID: 29709212
- PMCID: PMC9623596
- DOI: 10.1146/annurev-neuro-080317-061956
Memory Allocation: Mechanisms and Function
Abstract
Memories for events are thought to be represented in sparse, distributed neuronal ensembles (or engrams). In this article, we review how neurons are chosen to become part of a particular engram, via a process of neuronal allocation. Experiments in rodents indicate that eligible neurons compete for allocation to a given engram, with more excitable neurons winning this competition. Moreover, fluctuations in neuronal excitability determine how engrams interact, promoting either memory integration (via coallocation to overlapping engrams) or separation (via disallocation to nonoverlapping engrams). In parallel with rodent studies, recent findings in humans verify the importance of this memory integration process for linking memories that occur close in time or share related content. A deeper understanding of allocation promises to provide insights into the logic underlying how knowledge is normally organized in the brain and the disorders in which this process has gone awry.
Keywords: allocation; engram; fear; memory; neuronal excitability; reward.
Figures
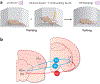

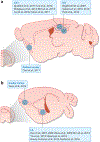


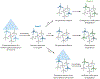
References
-
- Abraham WC, Bear MF. 1996. Metaplasticity: the plasticity of synaptic plasticity. Trends Neurosci. 19:126–30 - PubMed
-
- Alkon DL. 1984a. Calcium-mediated reduction of ionic currents: a biophysical memory trace. Science 226:1037–45 - PubMed
-
- Alkon DL. 1984b. Changes of membrane currents during learning. J. Exp. Biol 112:95–112 - PubMed
-
- Alkon DL, Lederhendler I, Shoukimas JJ. 1982. Primary changes of membrane currents during retention of associative learning. Science 215:693–95 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

