A randomized, double-blind, placebo-controlled Phase III trial of duloxetine in Japanese patients with knee pain due to osteoarthritis
- PMID: 29713194
- PMCID: PMC5912377
- DOI: 10.2147/JPR.S164128
A randomized, double-blind, placebo-controlled Phase III trial of duloxetine in Japanese patients with knee pain due to osteoarthritis
Abstract
Purpose: To examine the efficacy and safety of duloxetine in Japanese patients with knee pain due to osteoarthritis.
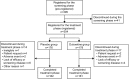
Patients and methods: Patients were randomized to receive duloxetine 60 mg/day or placebo for 14 weeks in a double-blind manner (ClinicalTrials.gov Identifier: NCT02248480). The primary efficacy endpoint was mean change in Brief Pain Inventory pain severity (BPI-Severity) average pain. Secondary endpoints included improvement in other BPI-Severity scales, Patient Global Impression of Improvement, Clinical Global Impressions of Severity, health-related quality of life (HRQoL) scales, range of motion of the knee joint, safety and tolerability, and structural changes on X-ray images.
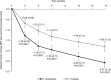
Results: Of the 354 randomized patients, 161 in the duloxetine group and 162 in the placebo group completed the study. BPI-Severity average pain improved significantly with duloxetine vs. placebo (-2.57 vs. -1.80; adjusted mean difference: -0.77; 95% CI: -1.11 to -0.43; P<0.0001). Secondary efficacy endpoints and most HRQoL scales showed greater improvements in the duloxetine group than the placebo group. Adverse events observed in ≥5% of patients that were more frequent in the duloxetine than placebo group were somnolence, constipation, dry mouth, nausea, malaise, and decreased appetite. There were no marked changes in range of motion of the knee joint (efficacy), X-ray images, or Kellgren-Lawrence grade (safety) in either group.
Conclusion: Duloxetine reduced pain and improved function in patients with knee osteoarthritis, without causing X-ray abnormalities or altered knee joint mobility. Reduced pain was associated with improved HRQoL. Adverse events were consistent with duloxetine's known safety profile.
Keywords: double-blind; duloxetine; osteoarthritis; placebo-controlled trial; randomized.
Figures


Similar articles
-
Safety and efficacy of duloxetine in Japanese patients with chronic knee pain due to osteoarthritis: an open-label, long-term, Phase III extension study.J Pain Res. 2018 Jul 31;11:1391-1403. doi: 10.2147/JPR.S171395. eCollection 2018. J Pain Res. 2018. PMID: 30104894 Free PMC article.
-
Randomized, Double-blind, Placebo-controlled Phase III Trial of Duloxetine Monotherapy in Japanese Patients With Chronic Low Back Pain.Spine (Phila Pa 1976). 2016 Nov 15;41(22):1709-1717. doi: 10.1097/BRS.0000000000001707. Spine (Phila Pa 1976). 2016. PMID: 27831985 Free PMC article. Clinical Trial.
-
Duloxetine added to oral nonsteroidal anti-inflammatory drugs for treatment of knee pain due to osteoarthritis: results of a randomized, double-blind, placebo-controlled trial.Curr Med Res Opin. 2011 Dec;27(12):2361-72. doi: 10.1185/03007995.2011.633502. Epub 2011 Nov 9. Curr Med Res Opin. 2011. PMID: 22017192 Clinical Trial.
-
Duloxetine: a review of its use in the treatment of generalized anxiety disorder.CNS Drugs. 2009;23(6):523-41. doi: 10.2165/00023210-200923060-00006. CNS Drugs. 2009. PMID: 19480470 Review.
-
An Updated Systematic Review and Meta-analysis of Duloxetine for Knee Osteoarthritis Pain.Clin J Pain. 2021 Nov 1;37(11):852-862. doi: 10.1097/AJP.0000000000000975. Clin J Pain. 2021. PMID: 34483232 Free PMC article.
Cited by
-
Does sex affect the efficacy of systemic pharmacological treatments of pain in knee osteoarthritis? A systematic review.Osteoarthr Cartil Open. 2024 Feb 10;6(1):100438. doi: 10.1016/j.ocarto.2024.100438. eCollection 2024 Mar. Osteoarthr Cartil Open. 2024. PMID: 38375468 Free PMC article. Review.
-
Clinical meaningfulness of duloxetine's effect in Chinese patients with chronic pain due to osteoarthritis: post hoc analyses of a phase 3 randomized trial.Open Access Rheumatol. 2019 Mar 21;11:67-76. doi: 10.2147/OARRR.S193044. eCollection 2019. Open Access Rheumatol. 2019. PMID: 30962729 Free PMC article.
-
Diet and companionship modulate pain via a serotonergic mechanism.Sci Rep. 2021 Feb 1;11(1):2330. doi: 10.1038/s41598-021-81654-1. Sci Rep. 2021. PMID: 33526805 Free PMC article.
-
Response to duloxetine in patients with knee pain due to osteoarthritis: an exploratory post hoc analysis of a Japanese Phase III randomized study.J Pain Res. 2018 Oct 26;11:2603-2616. doi: 10.2147/JPR.S176036. eCollection 2018. J Pain Res. 2018. PMID: 30464579 Free PMC article.
-
Relationship Between Pain Reduction and Improvement in Health-Related Quality of Life in Patients with Knee Pain Due to Osteoarthritis Receiving Duloxetine: Exploratory Post Hoc Analysis of a Japanese Phase 3 Randomized Study.J Pain Res. 2020 Jan 20;13:181-191. doi: 10.2147/JPR.S211072. eCollection 2020. J Pain Res. 2020. PMID: 32021407 Free PMC article.
References
-
- Chappell AS, Ossanna MJ, Liu-Seifert H, et al. Duloxetine, a centrally acting analgesic, in the treatment of patients with osteoarthritis knee pain: a 13-week, randomized, placebo-controlled trial. Pain. 2009;146(3):253–260. - PubMed
-
- Chappell AS, Desaiah D, Liu-Seifert H, et al. A double-blind, randomized, placebo-controlled study of the efficacy and safety of duloxetine for the treatment of chronic pain due to osteoarthritis of the knee. Pain Pract. 2011;11(1):33–41. - PubMed
-
- GBD 2015 Disease and Injury Incidence and Prevalence Collaborators Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388(10053):1545–1602. - PMC - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

