Dehydroepiandrosterone Antagonizes Pain Stress-Induced Suppression of Testosterone Production in Male Rats
- PMID: 29713278
- PMCID: PMC5911460
- DOI: 10.3389/fphar.2018.00322
Dehydroepiandrosterone Antagonizes Pain Stress-Induced Suppression of Testosterone Production in Male Rats
Abstract
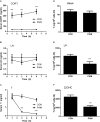
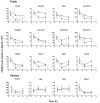
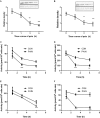
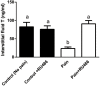
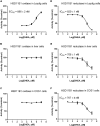
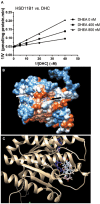
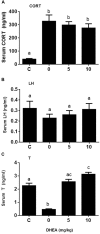
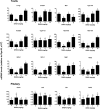
Background: Leydig cells secrete the steroid hormone, testosterone, which is essential for male fertility and reproductive health. Stress increases the secretion of glucocorticoid [corticosterone, (CORT) in rats] that decreases circulating testosterone levels in part through a direct action on its receptors in Leydig cells. Intratesticular CORT level is dependent on oxidative inactivation of CORT by 11β-hydroxysteroid dehydrogenase 1 (HSD11B1) in rat Leydig cells. Pain may cause the stress, thus affecting testosterone production in Leydig cells. Methods: Adult male Sprague-Dawley rats orally received vehicle control or 5 or 10 mg/kg dehydroepiandrosterone (DHEA) 0.5 h before being subjected to pain stimulation for 1, 3, and 6 h. In the present study, we investigated the time-course changes of steroidogenic gene expression levels after acute pain-induced stress in rats and the possible mechanism of DHEA that prevented it. Plasma CORT, luteinizing hormone (LH), and testosterone (T) levels were measured, and Leydig cell gene expression levels were determined. The direct regulation of HSD11B1 catalytic direction by DHEA was detected in purified rat Leydig, liver, and rat Hsd11b1-transfected COS1 cells. Results: Plasma CORT levels were significantly increased at hour 1, 3, and 6 during the pain stimulation, while plasma T levels were significantly decreased starting at hour 3 and 6. Pain-induced stress also decreased Star, Hsd3b1, and Cyp17a1 expression levels at hour 3. When 5 and 10 mg/kg DHEA were orally administered to rats 0.5 h before starting pain stimulation, DHEA prevented pain-mediated decrease in plasma T levels and the expression of Star, Hsd3b1, and Cyp17a1 without affecting plasma CORT levels. DHEA was found to modulate HSD11B1 activities by increasing its oxidative activity and decreasing its reductive activity, thus decreasing the intracellular CORT levels in Leydig cells. Conclusion: Stress induced by acute pain can inhibit Leydig cell T production by upregulation of corticosterone. DHEA can prevent the negative effects of excessive corticosterone by modulating HSD11B1 activity.
Keywords: 11β-hydroxysteroid dehydrogenase 1; DHEA; Leydig cell; acute stress; corticosterone; pain; steroidogenic enzymes.
Figures
Similar articles
-
Time-course changes of steroidogenic gene expression and steroidogenesis of rat Leydig cells after acute immobilization stress.Int J Mol Sci. 2014 Nov 14;15(11):21028-44. doi: 10.3390/ijms151121028. Int J Mol Sci. 2014. PMID: 25405735 Free PMC article.
-
Inhibition of 11beta-hydroxysteroid dehydrogenase enzymatic activities by glycyrrhetinic acid in vivo supports direct glucocorticoid-mediated suppression of steroidogenesis in Leydig cells.J Androl. 2008 May-Jun;29(3):345-51. doi: 10.2164/jandrol.107.004242. Epub 2008 Jan 9. J Androl. 2008. PMID: 18187395
-
Luteinizing hormone induces expression of 11beta-hydroxysteroid dehydrogenase type 2 in rat Leydig cells.Reprod Biol Endocrinol. 2009 May 4;7:39. doi: 10.1186/1477-7827-7-39. Reprod Biol Endocrinol. 2009. PMID: 19409113 Free PMC article.
-
Stress hormone and male reproductive function.Cell Tissue Res. 2005 Oct;322(1):147-53. doi: 10.1007/s00441-005-0006-2. Epub 2005 Nov 3. Cell Tissue Res. 2005. PMID: 16079965 Review.
-
The cross talk of adrenal and Leydig cell steroids in Leydig cells.J Steroid Biochem Mol Biol. 2019 Sep;192:105386. doi: 10.1016/j.jsbmb.2019.105386. Epub 2019 May 29. J Steroid Biochem Mol Biol. 2019. PMID: 31152782 Review.
Cited by
-
Association between the prevalence rates of circadian syndrome and testosterone deficiency in US males: data from NHANES (2011-2016).Front Nutr. 2023 May 9;10:1137668. doi: 10.3389/fnut.2023.1137668. eCollection 2023. Front Nutr. 2023. PMID: 37229470 Free PMC article.
-
Neonatal corticosterone administration increases p27-positive Sertoli cell number and decreases Sertoli cell number in the testes of mice at prepuberty.Sci Rep. 2022 Nov 12;12(1):19402. doi: 10.1038/s41598-022-23695-8. Sci Rep. 2022. PMID: 36371473 Free PMC article.
References
-
- Agarwal A. K., Monder C., Eckstein B., White P. C. (1989). Cloning and expression of rat cDNA encoding corticosteroid 11β-dehydrogenase. J. Biol. Chem. 264 18939–18943. - PubMed
-
- Akibami M. A., Mann D. R. (1996). Mechanism of stress-induced attenuation of the testicular response to gonadotropin: possible involvement of testicular opioids, a pertussis toxin-sensitive G-protein, and phosphodiesterase. J. Androl. 17 10–16. - PubMed
-
- Akingbemi B. T., Sottas C. M., Koulova A. I., Klinefelter G. R., Hardy M. P. (2004). Inhibition of testicular steroidogenesis by the xenoestrogen bisphenol A is associated with reduced pituitary luteinizing hormone secretion and decreased steroidogenic enzyme gene expression in rat Leydig cells. Endocrinology 145 592–603. 10.1210/en.2003-1174 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

