Anti-CTLA-4 antibodies in cancer immunotherapy: selective depletion of intratumoral regulatory T cells or checkpoint blockade?
- PMID: 29713453
- PMCID: PMC5907314
- DOI: 10.1186/s13578-018-0229-z
Anti-CTLA-4 antibodies in cancer immunotherapy: selective depletion of intratumoral regulatory T cells or checkpoint blockade?
Abstract
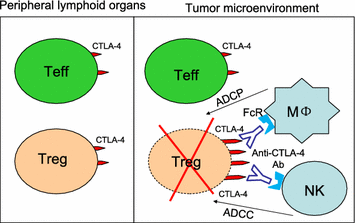
Antibodies to human CTLA-4 have been shown to induce long-lasting protection against melanoma. It is assumed that these antibodies cause tumor rejection by blocking negative signaling from the B7-CTLA-4 interactions to enhance priming of naïve T cells in the lymphoid organs. Recently, we reported that anti-CTLA-4 antibody Ipilimumab effectively induces tumor rejection in vivo although it blocks neither B7 transendocytosis by CTLA-4 nor CTLA-4 binding to immobilized or cell-associated B7. Using genetic model in which the anti-CTLA-4 antibodies are unable to engage more than 50% of CTLA-4, we demonstrated that saturating binding of CTLA-4 is not necessary for tumor rejection. Our results argue against B7-CTLA-4 blockade as the mechanism of action for the clinically effective Ipilimumab. Moreover, Ipilimumab induces tumor rejection even in the absence of de novo T cell priming in the lymphoid organs. Thus, our data are inconsistent with key provisions of the prevailing hypothesis on mechanism of action by anti-CTLA-4 antibodies. Furthermore, anti-CTLA-4 antibodies effectively induce depletion of regulatory T (Treg) cells in tumor microenvironment but not in the peripheral lymphoid organs, which is strictly dependent on Fc receptor on host cells. Based on these data and other recent publications on the subject, we propose that anti-human CTLA-4 antibodies induce tumor rejection by selective depletion of Tregs in the tumors rather than blockade of B7-CTLA-4 interaction in lymphoid organs.
Keywords: B7-CTLA-4 interaction; CTLA-4; Cancer immunotherapy; Ipilimumab; Regulatory T cells; Tumor microenvironment.
Figures

Similar articles
-
A reappraisal of CTLA-4 checkpoint blockade in cancer immunotherapy.Cell Res. 2018 Apr;28(4):416-432. doi: 10.1038/s41422-018-0011-0. Epub 2018 Feb 22. Cell Res. 2018. PMID: 29472691 Free PMC article.
-
Anti-CTLA-4 Immunotherapy Does Not Deplete FOXP3+ Regulatory T Cells (Tregs) in Human Cancers.Clin Cancer Res. 2019 Feb 15;25(4):1233-1238. doi: 10.1158/1078-0432.CCR-18-0762. Epub 2018 Jul 27. Clin Cancer Res. 2019. PMID: 30054281 Free PMC article.
-
Both intratumoral regulatory T cell depletion and CTLA-4 antagonism are required for maximum efficacy of anti-CTLA-4 antibodies.Proc Natl Acad Sci U S A. 2023 Aug;120(31):e2300895120. doi: 10.1073/pnas.2300895120. Epub 2023 Jul 24. Proc Natl Acad Sci U S A. 2023. PMID: 37487077 Free PMC article.
-
Addressing the Elephant in the Immunotherapy Room: Effector T-Cell Priming versus Depletion of Regulatory T-Cells by Anti-CTLA-4 Therapy.Cancers (Basel). 2022 Mar 20;14(6):1580. doi: 10.3390/cancers14061580. Cancers (Basel). 2022. PMID: 35326731 Free PMC article. Review.
-
Targeting cytotoxic T-lymphocyte antigen-4 (CTLA-4): a novel strategy for the treatment of melanoma and other malignancies.Cancer. 2007 Dec 15;110(12):2614-27. doi: 10.1002/cncr.23086. Cancer. 2007. PMID: 18000991 Review.
Cited by
-
Mechanism- and Immune Landscape-Based Ranking of Therapeutic Responsiveness of 22 Major Human Cancers to Next Generation Anti-CTLA-4 Antibodies.Cancers (Basel). 2020 Jan 24;12(2):284. doi: 10.3390/cancers12020284. Cancers (Basel). 2020. PMID: 31991588 Free PMC article.
-
Targeting the tumor microenvironment in B-cell lymphoma: challenges and opportunities.J Hematol Oncol. 2021 Aug 17;14(1):125. doi: 10.1186/s13045-021-01134-x. J Hematol Oncol. 2021. PMID: 34404434 Free PMC article. Review.
-
Disease Etiology Impact on Outcomes of Hepatocellular Carcinoma Patients Treated with Atezolizumab plus Bevacizumab: A Real-World, Multicenter Study.Liver Cancer. 2024 Apr 10;13(5):522-536. doi: 10.1159/000537915. eCollection 2024 Oct. Liver Cancer. 2024. PMID: 39296620 Free PMC article.
-
Phase II Study of Ipilimumab in Men With Metastatic Prostate Cancer With an Incomplete Response to Androgen Deprivation Therapy.Front Oncol. 2020 Aug 7;10:1381. doi: 10.3389/fonc.2020.01381. eCollection 2020. Front Oncol. 2020. PMID: 32850444 Free PMC article.
-
Treatment of recurrent ovarian germ cell tumours: Is there a role for immune checkpoint inhibitors?Gynecol Oncol Rep. 2024 Sep 19;56:101502. doi: 10.1016/j.gore.2024.101502. eCollection 2024 Dec. Gynecol Oncol Rep. 2024. PMID: 39386925 Free PMC article. Review.
References
-
- Lute KD, May KF, Jr, Lu P, Zhang H, Kocak E, Mosinger B, Wolford C, Phillips G, Caligiuri MA, Zheng P, Liu Y. Human CTLA4 knock-in mice unravel the quantitative link between tumor immunity and autoimmunity induced by anti-CTLA-4 antibodies. Blood. 2005;106:3127–3133. doi: 10.1182/blood-2005-06-2298. - DOI - PMC - PubMed
-
- May KF, Jr, Roychowdhury S, Bhatt D, Kocak E, Bai XF, Liu JQ, Ferketich AK, Martin EW, Jr, Caligiuri MA, Zheng P, Liu Y. Anti-human CTLA-4 monoclonal antibody promotes T-cell expansion and immunity in a hu-PBL-SCID model: a new method for preclinical screening of costimulatory monoclonal antibodies. Blood. 2005;105:1114–1120. doi: 10.1182/blood-2004-07-2561. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

