Stress and stem cells: adult Muse cells tolerate extensive genotoxic stimuli better than mesenchymal stromal cells
- PMID: 29721206
- PMCID: PMC5922400
- DOI: 10.18632/oncotarget.25039
Stress and stem cells: adult Muse cells tolerate extensive genotoxic stimuli better than mesenchymal stromal cells
Abstract
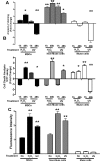
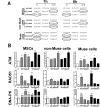
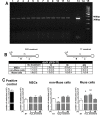
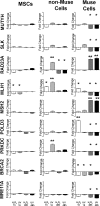
Mesenchymal stromal cells (MSCs) are not a homogenous population but comprehend several cell types, such as stem cells, progenitor cells, fibroblasts, and other types of cells. Among these is a population of pluripotent stem cells, which represent around 1-3% of MSCs. These cells, named multilineage-differentiating stress enduring (Muse) cells, are stress-tolerant cells. Stem cells may undergo several rounds of intrinsic and extrinsic stresses due to their long life and must have a robust and effective DNA damage checkpoint and DNA repair mechanism, which, following a genotoxic episode, promote the complete recovery of cells rather than triggering senescence and/or apoptosis. We evaluated how Muse cells can cope with DNA damaging stress in comparison with MSCs. We found that Muse cells were resistant to chemical and physical genotoxic stresses better than non-Muse cells. Indeed, the level of senescence and apoptosis was lower in Muse cells. Our results proved that the DNA damage repair system (DDR) was properly activated following injury in Muse cells. While in non-Muse cells some anomalies may have occurred because, in some cases, the activation of the DDR persisted by 48 hr post damage, in others no activation took place. In Muse cells, the non-homologous end joining (NHEJ) enzymatic activity increases compared to other cells, while single-strand repair activity (NER, BER) does not. In conclusion, the high ability of Muse cells to cope with genotoxic stress is related to their quick and efficient sensing of DNA damage and activation of DNA repair systems.
Keywords: apoptosis; mesenchymal stem cells; senescence.
Conflict of interest statement
CONFLICTS OF INTEREST None.
Figures

References
-
- Galderisi U, Giordano A. The gap between the physiological and therapeutic roles of mesenchymal stem cells. Med Res Rev. 2014;34:1100–26. https://doi.org/10.1002/med.21322. - DOI - PubMed
-
- Wakao S, Kuroda Y, Ogura F, Shigemoto T, Dezawa M. Regenerative Effects of Mesenchymal Stem Cells: Contribution of Muse Cells, a Novel Pluripotent Stem Cell Type that Resides in Mesenchymal Cells. Cells. 2012;1:1045–60. https://doi.org/10.3390/cells1041045. - DOI - PMC - PubMed
-
- Wakao S, Kitada M, Kuroda Y, Shigemoto T, Matsuse D, Akashi H, Tanimura Y, Tsuchiyama K, Kikuchi T, Goda M, Nakahata T, Fujiyoshi Y, Dezawa M. Multilineage-differentiating stress-enduring (Muse) cells are a primary source of induced pluripotent stem cells in human fibroblasts. Proc Natl Acad Sci U S A. 2011;108:9875–80. https://doi.org/10.1073/pnas.1100816108. - DOI - PMC - PubMed
-
- Dezawa M. Muse cells provide the pluripotency of mesenchymal stem cells: direct contribution of Muse cells to tissue regeneration. Cell Transplant. 2016;25:849–61. https://doi.org/10.3727/096368916X690881. - DOI - PubMed
-
- Tsuchiyama K, Wakao S, Kuroda Y, Ogura F, Nojima M, Sawaya N, Yamasaki K, Aiba S, Dezawa M. Functional melanocytes are readily reprogrammable from multilineage-differentiating stress-enduring (muse) cells, distinct stem cells in human fibroblasts. J Invest Dermatol. 2013;133:2425–35. https://doi.org/10.1038/jid.2013.172. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

