Rapid Turnover and High Production Rate of Myeloid Cells in Adult Rhesus Macaques with Compensations during Aging
- PMID: 29728510
- PMCID: PMC6263173
- DOI: 10.4049/jimmunol.1800207
Rapid Turnover and High Production Rate of Myeloid Cells in Adult Rhesus Macaques with Compensations during Aging
Abstract
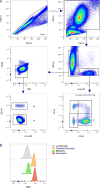
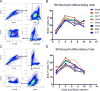
Neutrophils, basophils, and monocytes are continuously produced in bone marrow via myelopoiesis, circulate in blood, and are eventually removed from circulation to maintain homeostasis. To quantitate the kinetics of myeloid cell movement during homeostasis, we applied 5-bromo-2'-deoxyuridine pulse labeling in healthy rhesus macaques (Macaca mulatta) followed by hematology and flow cytometry analyses. Results were applied to a mathematical model, and the blood circulating half-life and daily production, respectively, of each cell type from macaques aged 5-10 y old were calculated for neutrophils (1.63 ± 0.16 d, 1.42 × 109 cells/l/d), basophils (1.78 ± 0.30 d, 5.89 × 106 cells/l/d), and CD14+CD16- classical monocytes (1.01 ± 0.15 d, 3.09 × 108 cells/l/d). Classical monocytes were released into the blood circulation as early as 1 d after dividing, whereas neutrophils remained in bone marrow 4-5 d before being released. Among granulocytes, neutrophils and basophils exhibited distinct kinetics in bone marrow maturation time and blood circulation. With increasing chronological age, there was a significant decrease in daily production of neutrophils and basophils, but the half-life of these granulocytes remained unchanged between 3 and 19 y of age. In contrast, daily production of classical monocytes remained stable through 19 y of age but exhibited a significant decline in half-life. These results demonstrated relatively short half-lives and continuous replenishment of neutrophils, basophils, and classical monocytes during homeostasis in adult rhesus macaques with compensations observed during increasing chronological age.
Copyright © 2018 by The American Association of Immunologists, Inc.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
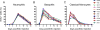
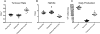
Similar articles
-
Differentiation Kinetics of Blood Monocytes and Dendritic Cells in Macaques: Insights to Understanding Human Myeloid Cell Development.J Immunol. 2015 Aug 15;195(4):1774-81. doi: 10.4049/jimmunol.1500522. Epub 2015 Jul 15. J Immunol. 2015. PMID: 26179903 Free PMC article.
-
Declining neutrophil production despite increasing G-CSF levels is associated with chronic inflammation in elderly rhesus macaques.J Leukoc Biol. 2021 Jun;109(6):1033-1043. doi: 10.1002/JLB.1HI1120-779R. Epub 2021 May 11. J Leukoc Biol. 2021. PMID: 33974319 Free PMC article.
-
Estimation of kinetic parameters of neutrophilic, eosinophilic, and basophilic granulocytes in human blood.Blut. 1979 Jul;39(1):27-38. doi: 10.1007/BF01008072. Blut. 1979. PMID: 223692
-
The twilight zone: plasticity and mixed ontogeny of neutrophil and eosinophil granulocyte subsets.Semin Immunopathol. 2021 Jun;43(3):337-346. doi: 10.1007/s00281-021-00862-z. Epub 2021 May 19. Semin Immunopathol. 2021. PMID: 34009400 Free PMC article. Review.
-
What's your age again? Determination of human neutrophil half-lives revisited.J Leukoc Biol. 2013 Oct;94(4):595-601. doi: 10.1189/jlb.1112571. Epub 2013 Apr 26. J Leukoc Biol. 2013. PMID: 23625199 Review.
Cited by
-
Monocyte subsets display age-dependent alterations at fasting and undergo non-age-dependent changes following consumption of a meal.Immun Ageing. 2022 Sep 14;19(1):41. doi: 10.1186/s12979-022-00297-6. Immun Ageing. 2022. PMID: 36104734 Free PMC article.
-
Monocyte Polarization is Altered by Total-Body Irradiation in Male Rhesus Macaques: Implications for Delayed Effects of Acute Radiation Exposure.Radiat Res. 2019 Aug;192(2):121-134. doi: 10.1667/RR15310.1. Epub 2019 Jun 4. Radiat Res. 2019. PMID: 31161966 Free PMC article.
-
Interconnections between Inflammageing and Immunosenescence during Ageing.Cells. 2022 Jan 21;11(3):359. doi: 10.3390/cells11030359. Cells. 2022. PMID: 35159168 Free PMC article. Review.
-
The Host Cellular Immune Response to Infection by Campylobacter Spp. and Its Role in Disease.Infect Immun. 2021 Jul 15;89(8):e0011621. doi: 10.1128/IAI.00116-21. Epub 2021 Jul 15. Infect Immun. 2021. PMID: 34031129 Free PMC article. Review.
-
Monocytes in HIV and SIV Infection and Aging: Implications for Inflamm-Aging and Accelerated Aging.Viruses. 2022 Feb 17;14(2):409. doi: 10.3390/v14020409. Viruses. 2022. PMID: 35216002 Free PMC article. Review.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

