Development of nanoemulsion for efficient brain parenteral delivery of cefuroxime: designs, characterizations, and pharmacokinetics
- PMID: 29731632
- PMCID: PMC5927357
- DOI: 10.2147/IJN.S151788
Development of nanoemulsion for efficient brain parenteral delivery of cefuroxime: designs, characterizations, and pharmacokinetics
Abstract
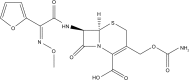
Background and aim: Drugs that are effective against diseases in the central nervous system and reach the brain via blood must pass through the blood-brain barrier (BBB), a unique interface that protects against potential harmful molecules. This presents a major challenge in neuro-drug delivery. This study attempts to fabricate the cefuroxime-loaded nanoemulsion (CLN) to increase drug penetration into the brain when parenterally administered.
Methods: The nanoemulsions were formulated using a high-pressure homogenization technique and were characterized for their physicochemical properties.
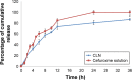
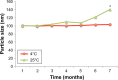
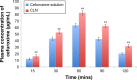
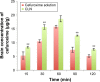
Results: The characterizations revealed a particle size of 100.32±0.75 nm, polydispersity index of 0.18±0.01, zeta potential of -46.9±1.39 mV, viscosity of 1.24±0.34 cps, and osmolality of 285.33±0.58 mOsm/kg, indicating that the nanoemulsion has compatibility for parenteral application. CLN was physicochemically stable within 6 months of storage at 4°C, and the transmission electron microscopy revealed that the CLN droplets were almost spherical in shape. The in vitro release of CLN profile followed a sustained release pattern. The pharmacokinetic profile of CLN showed a significantly higher Cmax, area under the curve (AUC)0-t , prolonged half-life, and lower total plasma clearance, indicating that the systemic concentration of cefuroxime was higher in CLN-treated rats as compared to cefuroxime-free treated rats. A similar profile was obtained for the biodistribution of cefuroxime in the brain, in which CLN showed a significantly higher Cmax, AUC0-t , prolonged half-life, and lower clearance as compared to free cefuroxime solution.
Conclusion: Overall, CLN showed excellent physicochemical properties, fulfilled the requirements for parenteral administration, and presented improved in vivo pharmacokinetic profile, which reflected its practical approach to enhance cefuroxime delivery to the brain.
Keywords: blood–brain barrier; cefuroxime; drug delivery; parenteral nanoemulsion; pharmacokinetics.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures


Similar articles
-
Nanoemulsion-based Parenteral Drug Delivery System of Carbamazepine: Preparation, Characterization, Stability Evaluation and Blood-Brain Pharmacokinetics.Curr Drug Deliv. 2015;12(6):795-804. doi: 10.2174/1567201812666150901112544. Curr Drug Deliv. 2015. PMID: 26324229
-
Characterisation, in-vitro and in-vivo evaluation of valproic acid-loaded nanoemulsion for improved brain bioavailability.J Pharm Pharmacol. 2017 Nov;69(11):1447-1457. doi: 10.1111/jphp.12800. Epub 2017 Aug 15. J Pharm Pharmacol. 2017. PMID: 28809443
-
Engineering of Long-Term Stable Transparent Nanoemulsion Using High-Gravity Rotating Packed Bed for Oral Drug Delivery.Int J Nanomedicine. 2020 Apr 9;15:2391-2402. doi: 10.2147/IJN.S238788. eCollection 2020. Int J Nanomedicine. 2020. PMID: 32308390 Free PMC article.
-
Novel Approach for Transdermal Delivery of Rifampicin to Induce Synergistic Antimycobacterial Effects Against Cutaneous and Systemic Tuberculosis Using a Cationic Nanoemulsion Gel.Int J Nanomedicine. 2020 Feb 14;15:1073-1094. doi: 10.2147/IJN.S236277. eCollection 2020. Int J Nanomedicine. 2020. PMID: 32103956 Free PMC article.
-
A Novel Drug Delivery Carrier Comprised of Nimodipine Drug Solution and a Nanoemulsion: Preparation, Characterization, in vitro, and in vivo Studies.Int J Nanomedicine. 2020 Feb 18;15:1161-1172. doi: 10.2147/IJN.S226591. eCollection 2020. Int J Nanomedicine. 2020. PMID: 32110014 Free PMC article.
Cited by
-
Date Palm Extract (Phoenix dactylifera) PEGylated Nanoemulsion: Development, Optimization and Cytotoxicity Evaluation.Plants (Basel). 2021 Apr 9;10(4):735. doi: 10.3390/plants10040735. Plants (Basel). 2021. PMID: 33918742 Free PMC article.
-
Parenteral Nanoemulsion for Optimized Delivery of GL-II-73 to the Brain-Comparative In Vitro Blood-Brain Barrier and In Vivo Neuropharmacokinetic Evaluation.Pharmaceutics. 2025 Mar 10;17(3):354. doi: 10.3390/pharmaceutics17030354. Pharmaceutics. 2025. PMID: 40143018 Free PMC article.
-
Ultrasound-assisted production and optimization of mini-emulsions in a microfluidic chip in continuous-flow.Ultrason Sonochem. 2021 Jun;74:105556. doi: 10.1016/j.ultsonch.2021.105556. Epub 2021 Apr 15. Ultrason Sonochem. 2021. PMID: 33915482 Free PMC article.
-
Neurosurgery at the crossroads of immunology and nanotechnology. New reality in the COVID-19 pandemic.Adv Drug Deliv Rev. 2022 Feb;181:114033. doi: 10.1016/j.addr.2021.114033. Epub 2021 Nov 20. Adv Drug Deliv Rev. 2022. PMID: 34808227 Free PMC article. Review.
-
Oral administration of Huanglian-Houpo herbal nanoemulsion loading multiple phytochemicals for ulcerative colitis therapy in mice.Drug Deliv. 2023 Dec;30(1):2204207. doi: 10.1080/10717544.2023.2204207. Drug Deliv. 2023. PMID: 37139554 Free PMC article.
References
-
- Swartz MN, Dodge PR. Bacterial meningitis – a review of selected aspects. N Engl J Med. 1965;272(15):779–787. - PubMed
-
- Honda H, Warren DK. Central nervous system infections: meningitis and brain abscess. Infect Dis Clin North Am. 2009;23(3):609–623. - PubMed
-
- Manten A. Side effects of antibiotics. Vet Q. 1981;3(4):179–182. - PubMed
-
- Yılmaz Ç, Özcengiz G. Antibiotics: pharmacokinetics, toxicity, resistance and multidrug efflux pumps. Biochem Pharmacol. 2017;133:43–62. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

