Ceramide species are elevated in human breast cancer and are associated with less aggressiveness
- PMID: 29731990
- PMCID: PMC5929433
- DOI: 10.18632/oncotarget.24903
Ceramide species are elevated in human breast cancer and are associated with less aggressiveness
Abstract
Sphingolipids have emerged as key regulatory molecules in cancer cell survival and death. Although important roles of sphingolipids in breast cancer progression have been reported in experimental models, their roles in human patients are yet to be revealed. The aim of this study was to investigate the ceramide levels and its biosynthesis pathways in human breast cancer patients. Breast cancer, peri-tumor and normal breast tissue samples were collected from surgical specimens from a series of 44 patients with breast cancer. The amount of sphingolipid metabolites in the tissue were determined by mass spectrometry. The Cancer Genome Atlas was used to analyze gene expression related to the sphingolipid metabolism. Ceramide levels were higher in breast cancer tissue compared to both normal and peri-tumor breast tissue. Substrates and enzymes that generate ceramide were significantly increased in all three ceramide biosynthesis pathways in cancer. Further, higher levels of ceramide in breast cancer were associated with less aggressive cancer biology presented by Ki-67 index and nuclear grade of the cancer. Interestingly, patients with higher gene expressions of enzymes in the three major ceramide synthesis pathways showed significantly worse prognosis. This is the first study to reveal the clinical relevance of ceramide metabolism in breast cancer patients. We demonstrated that ceramide levels in breast cancer tissue were significantly higher than those in normal tissue, with activation of the three ceramide biosynthesis pathways. We also identified that ceramide levels have a significant association with aggressive phenotype and its enzymes have prognostic impact on breast cancer patients.
Keywords: breast cancer; ceramide; metabolism; prognosis; sphingolipid.
Conflict of interest statement
CONFLICTS OF INTEREST The authors have no conflicts of interest to disclose.
Figures




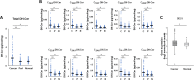


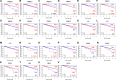
References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68:7–30. https://doi.org/10.3322/caac.21442 - DOI - PubMed
-
- Galadari S, Rahman A, Pallichankandy S, Thayyullathil F. Tumor suppressive functions of ceramide: evidence and mechanisms. Apoptosis. 2015;20:689–711. https://doi.org/10.1007/s10495-015-1109-1 - DOI - PubMed
-
- Spiegel S, Milstien S. Sphingosine-1-phosphate: an enigmatic signalling lipid. Nat Rev Mol Cell Biol. 2003;4:397–407. https://doi.org/10.1038/nrm1103 - DOI - PubMed
-
- Nagahashi M, Tsuchida J, Moro K, Hasegawa M, Tatsuda K, Woelfel IA, Takabe K, Wakai T. High levels of sphingolipids in human breast cancer. J Surg Res. 2016;204:435–44. https://doi.org/10.1016/j.jss.2016.05.022 - DOI - PMC - PubMed
-
- Takabe K, Kim RH, Allegood JC, Mitra P, Ramachandran S, Nagahashi M, Harikumar KB, Hait NC, Milstien S, Spiegel S. Estradiol induces export of sphingosine 1-phosphate from breast cancer cells via ABCC1 and ABCG2. J Biol Chem. 2010;285:10477–86. https://doi.org/10.1074/jbc.M109.064162 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

