Hepcidin Protects against Lethal Escherichia coli Sepsis in Mice Inoculated with Isolates from Septic Patients
- PMID: 29735522
- PMCID: PMC6013672
- DOI: 10.1128/IAI.00253-18
Hepcidin Protects against Lethal Escherichia coli Sepsis in Mice Inoculated with Isolates from Septic Patients
Abstract
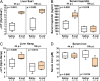
Iron is an essential micronutrient for most microbes and their hosts. Mammalian hosts respond to infection by inducing the iron-regulatory hormone hepcidin, which causes iron sequestration and a rapid decrease in the plasma and extracellular iron concentration (hypoferremia). Previous studies showed that hepcidin regulation of iron is essential for protection from infection-associated mortality with the siderophilic pathogens Yersinia enterocolitica and Vibrio vulnificus However, the evolutionary conservation of the hypoferremic response to infection suggests that not only rare siderophilic bacteria but also common pathogens may be targeted by this mechanism. We tested 10 clinical isolates of Escherichia coli from children with sepsis and found that both genetic iron overload (by hepcidin-1 knockout [HKO]) and iatrogenic iron overload (by intravenous iron) potentiated infection with 8 out of the 10 studied isolates: after peritoneal injection of E. coli, iron-loaded mice developed sepsis with 60% to 100% mortality within 24 h, while control wild-type mice suffered 0% mortality. Using one strain for more detailed study, we show that iron overload allows rapid bacterial multiplication and dissemination. We further found that the presence of non-transferrin-bound iron (NTBI) in the circulation is more important than total plasma or tissue iron in rendering mice susceptible to infection and mortality. Postinfection treatment of HKO mice with just two doses of the hepcidin agonist PR73 abolished NTBI and completely prevented sepsis-associated mortality. We demonstrate that the siderophilic phenotype extends to clinically common pathogens. The use of hepcidin agonists promises to be an effective early intervention in patients with infections and dysregulated iron metabolism.
Keywords: Escherichia coli; NTBI; hepcidin; infection; iron; sepsis.
Copyright © 2018 American Society for Microbiology.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

