The European Medicines Agency's approval of new medicines for type 2 diabetes
- PMID: 29740935
- PMCID: PMC6667915
- DOI: 10.1111/dom.13349
The European Medicines Agency's approval of new medicines for type 2 diabetes
Abstract
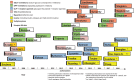
Since 2005, more than 40 new medicines for the treatment of type 2 diabetes have been introduced on the market. These consist of 15 new active substances establishing three new classes of non-insulin products, and several new or modified insulin products and combinations. The approval of these products in Europe is regulated via the centralized procedure at the European Medicines Agency. Demonstration of benefit with regard to improved glucose control remains the principal outcome required from confirmatory studies to demonstrate efficacy. For the majority of these new medicines approved since 2005, cardiovascular outcome trials have now been completed, and have invariably supported the cardiovascular safety of these products. In some of these trials additional important benefits have been observed, for instance, a reduction in major adverse cardiovascular events and improvement of renal outcome. The existing regulatory framework and the continuous adaption of regulatory requirements to emerging developments will continue to guide the approval of new products in the future.
Keywords: antidiabetic drug; drug development; systematic review.
© 2018 John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- OECD/EU . Health at a Glance: Europe 2016 – State of Health in the EU Cycle. Paris: OECD Publishing; 2016.
-
- Garg SK, Henry RR, Banks P, et al. Effects of Sotagliflozin added to insulin in patients with type 1 diabetes. N Engl J Med. 2017;377:2337‐2348. - PubMed
-
- Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycaemia in type 2 diabetes, 2015: a patient‐centred approach. Update to a position statement of the American Diabetes Association and the European Association for the Study of diabetes. Diabetologia. 2015;58:429‐442. - PubMed
-
- Nathan DM, Buse JB, Davidson MB, et al. Management of hyperglycaemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy. A consensus statement from the American Diabetes Association and the European Association for the Study of diabetes. Diabetologia. 2006;49:1711‐1721. - PubMed
-
- Deacon CF, Lebovitz HE. Comparative review of dipeptidyl peptidase‐4 inhibitors and sulphonylureas. Diabetes Obes Metab. 2016;18:333‐347. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical