Basal-like breast cancer engages tumor-supportive macrophages via secreted factors induced by extracellular S100A4
- PMID: 29741811
- PMCID: PMC6120223
- DOI: 10.1002/1878-0261.12319
Basal-like breast cancer engages tumor-supportive macrophages via secreted factors induced by extracellular S100A4
Abstract
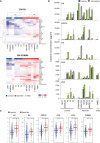
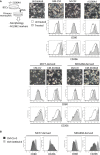
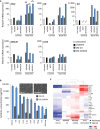
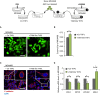
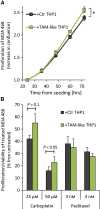
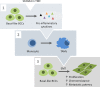
The tumor microenvironment (TME) may influence both cancer progression and therapeutic response. In breast cancer, particularly in the aggressive triple-negative/basal-like subgroup, patient outcome is strongly associated with the tumor's inflammatory profile. Tumor-associated macrophages (TAMs) are among the most abundant immune cells in the TME, shown to be linked to poor prognosis and therapeutic resistance. In this study, we investigated the effect of the metastasis- and inflammation-associated microenvironmental factor S100A4 on breast cancer cells (BCCs) of different subtypes and explored their further interactions with myeloid cells. We demonstrated that extracellular S100A4 activates BCCs, particularly the basal-like subtype, to elevate secretion of pro-inflammatory cytokines. The secreted factors promoted conversion of monocytes to TAM-like cells that exhibited protumorigenic activities: stimulated epithelial-mesenchymal transition, proliferation, chemoresistance, and motility in cancer cells. In conclusion, we have shown that extracellular S100A4 instigates a tumor-supportive microenvironment, involving a network of cytokines and TAM-like cells, which was particularly characteristic for basal-like BCCs and potentiated their aggressive properties. The S100A4-BCC-TAM interaction cascade could be an important contributor to the aggressive behavior of this subtype and should be further explored for therapeutic targeting.
Keywords: S100A4; breast cancer; cytokines; tumor microenvironment; tumor-associated macrophages; tumor-stroma interactions.
© 2018 The Authors. Published by FEBS Press and John Wiley & Sons Ltd.
Figures



References
-
- Benoy IH, Salgado R, Van Dam P, Geboers K, Van Marck E, Scharpe S, Vermeulen PB and Dirix LY (2004) Increased serum interleukin‐8 in patients with early and metastatic breast cancer correlates with early dissemination and survival. Clin Cancer Res 10, 7157–7162. - PubMed
-
- Berge G, Pettersen S, Grotterod I, Bettum IJ, Boye K and Maelandsmo GM (2011) Osteopontin – an important downstream effector of S100A4‐mediated invasion and metastasis. Int J Cancer 129, 780–790. - PubMed
-
- Bettum IJ, Vasiliauskaite K, Nygaard V, Clancy T, Pettersen SJ, Tenstad E, Maelandsmo GM and Prasmickaite L (2014) Metastasis‐associated protein S100A4 induces a network of inflammatory cytokines that activate stromal cells to acquire pro‐tumorigenic properties. Cancer Lett 344, 28–39. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

