Exploratory Application of Neuropharmacometabolomics in Severe Childhood Traumatic Brain Injury
- PMID: 29742587
- PMCID: PMC6095742
- DOI: 10.1097/CCM.0000000000003203
Exploratory Application of Neuropharmacometabolomics in Severe Childhood Traumatic Brain Injury
Abstract
Objectives: To employ metabolomics-based pathway and network analyses to evaluate the cerebrospinal fluid metabolome after severe traumatic brain injury in children and the capacity of combination therapy with probenecid and N-acetylcysteine to impact glutathione-related and other pathways and networks, relative to placebo treatment.
Design: Analysis of cerebrospinal fluid obtained from children enrolled in an Institutional Review Board-approved, randomized, placebo-controlled trial of a combination of probenecid and N-acetylcysteine after severe traumatic brain injury (Trial Registration NCT01322009).
Setting: Thirty-six-bed PICU in a university-affiliated children's hospital.
Patients and subjects: Twelve children 2-18 years old after severe traumatic brain injury and five age-matched control subjects.
Intervention: Probenecid (25 mg/kg) and N-acetylcysteine (140 mg/kg) or placebo administered via naso/orogastric tube.
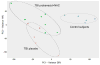
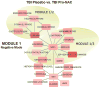
Measurements and main results: The cerebrospinal fluid metabolome was analyzed in samples from traumatic brain injury patients 24 hours after the first dose of drugs or placebo and control subjects. Feature detection, retention time, alignment, annotation, and principal component analysis and statistical analysis were conducted using XCMS-online. The software "mummichog" was used for pathway and network analyses. A two-component principal component analysis revealed clustering of each of the groups, with distinct metabolomics signatures. Several novel pathways with plausible mechanistic involvement in traumatic brain injury were identified. A combination of metabolomics and pathway/network analyses showed that seven glutathione-centered pathways and two networks were enriched in the cerebrospinal fluid of traumatic brain injury patients treated with probenecid and N-acetylcysteine versus placebo-treated patients. Several additional pathways/networks consisting of components that are known substrates of probenecid-inhibitable transporters were also identified, providing additional mechanistic validation.
Conclusions: This proof-of-concept neuropharmacometabolomics assessment reveals alterations in known and previously unidentified metabolic pathways and supports therapeutic target engagement of the combination of probenecid and N-acetylcysteine treatment after severe traumatic brain injury in children.
Figures



Comment in
-
Exposing and Engaging Unknown Unknowns in Traumatic Brain Injury.Crit Care Med. 2018 Sep;46(9):1556-1557. doi: 10.1097/CCM.0000000000003239. Crit Care Med. 2018. PMID: 30113376 No abstract available.
References
-
- Kaddurah-Daouk R, Krishnan KR. Metabolomics: a global biochemical approach to the study of central nervous system diseases. Neuropsychopharmacology. 2009;34(1):173–186. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

