Pauses in Cholinergic Interneuron Activity Are Driven by Excitatory Input and Delayed Rectification, with Dopamine Modulation
- PMID: 29754751
- PMCID: PMC5993868
- DOI: 10.1016/j.neuron.2018.04.027
Pauses in Cholinergic Interneuron Activity Are Driven by Excitatory Input and Delayed Rectification, with Dopamine Modulation
Abstract
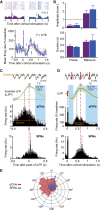
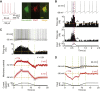
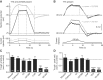
Cholinergic interneurons (ChIs) of the striatum pause their firing in response to salient stimuli and conditioned stimuli after learning. Several different mechanisms for pause generation have been proposed, but a unifying basis has not previously emerged. Here, using in vivo and ex vivo recordings in rat and mouse brain and a computational model, we show that ChI pauses are driven by withdrawal of excitatory inputs to striatum and result from a delayed rectifier potassium current (IKr) in concert with local neuromodulation. The IKr is sensitive to Kv7.2/7.3 blocker XE-991 and enables ChIs to report changes in input, to pause on excitatory input recession, and to scale pauses with input strength, in keeping with pause acquisition during learning. We also show that although dopamine can hyperpolarize ChIs directly, its augmentation of pauses is best explained by strengthening excitatory inputs. These findings provide a basis to understand pause generation in striatal ChIs. VIDEO ABSTRACT.
Keywords: basal ganglia; cholinergic interneuron; corticostriatal; delayed rectification; dopamine; excitatory input; nigrostriatal; pause response; striatum; thalamostriatal.
Copyright © 2018 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures

References
-
- Aosaki T., Graybiel A.M., Kimura M. Effect of the nigrostriatal dopamine system on acquired neural responses in the striatum of behaving monkeys. Science. 1994;265:412–415. - PubMed
-
- Aosaki T., Kimura M., Graybiel A.M. Temporal and spatial characteristics of tonically active neurons of the primate’s striatum. J. Neurophysiol. 1995;73:1234–1252. - PubMed
-
- Apicella P. Leading tonically active neurons of the striatum from reward detection to context recognition. Trends Neurosci. 2007;30:299–306. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

