Modulation of Receptor Protein Tyrosine Phosphatase Sigma Increases Chondroitin Sulfate Proteoglycan Degradation through Cathepsin B Secretion to Enhance Axon Outgrowth
- PMID: 29760175
- PMCID: PMC5990985
- DOI: 10.1523/JNEUROSCI.3214-17.2018
Modulation of Receptor Protein Tyrosine Phosphatase Sigma Increases Chondroitin Sulfate Proteoglycan Degradation through Cathepsin B Secretion to Enhance Axon Outgrowth
Abstract
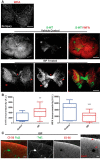
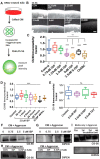
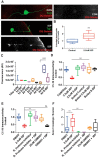
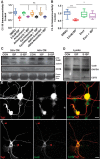
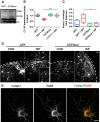
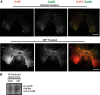
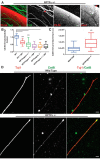
Severed axon tips reform growth cones following spinal cord injury that fail to regenerate, in part, because they become embedded within an inhibitory extracellular matrix. Chondroitin sulfate proteoglycans (CSPGs) are the major axon inhibitory matrix component that is increased within the lesion scar and in perineuronal nets around deafferented neurons. We have recently developed a novel peptide modulator (intracellular sigma peptide) of the cognate receptor of CSPGs, protein tyrosine phosphatase σ (RPTPσ), which has been shown to markedly improve sensorimotor function, micturition, and coordinated locomotor behavior in spinal cord contused rats. However, the mechanism(s) underlying how modulation of RPTPσ mediates axon outgrowth through inhibitory CSPGs remain unclear. Here, we describe how intracellular sigma peptide modulation of RPTPσ induces enhanced protease Cathepsin B activity. Using DRG neurons from female Sprague Dawley rats cultured on an aggrecan/laminin spot assay and a combination of biochemical techniques, we provide evidence suggesting that modulation of RPTPσ regulates secretion of proteases that, in turn, relieves CSPG inhibition through its digestion to allow axon migration though proteoglycan barriers. Understanding the mechanisms underlying RPTPσ modulation elucidates how axon regeneration is impaired by proteoglycans but can then be facilitated following injury.SIGNIFICANCE STATEMENT Following spinal cord injury, chondroitin sulfate proteoglycans (CSPGs) upregulate and potently inhibit axon regeneration and functional recovery. Protein tyrosine phosphatase σ (RPTPσ) has been identified as a critical cognate receptor of CSPGs. We have previously characterized a synthetic peptide (intracellular sigma peptide) that targets the regulatory intracellular domain of the receptor to allow axons to regenerate despite the presence of CSPGs. Here, we have found that one important mechanism by which peptide modulation of the receptor enhances axon outgrowth is through secretion of a protease, Cathepsin B, which enables digestion of CSPGs. This work links protease secretion to the CSPG receptor RPTPσ for the first time with implications for understanding the molecular mechanisms underlying neural regeneration and plasticity.
Keywords: CSPG; DRG; RPTPσ; axon regeneration; cathepsin B; protease.
Copyright © 2018 the authors 0270-6474/18/385399-16$15.00/0.
Figures

Similar articles
-
A new role for RPTPsigma in spinal cord injury: signaling chondroitin sulfate proteoglycan inhibition.Sci Signal. 2010 Feb 23;3(110):pe6. doi: 10.1126/scisignal.3110pe6. Sci Signal. 2010. PMID: 20179269
-
Chondroitin sulfate proteoglycans inhibit oligodendrocyte myelination through PTPσ.Exp Neurol. 2013 Sep;247:113-21. doi: 10.1016/j.expneurol.2013.04.003. Epub 2013 Apr 12. Exp Neurol. 2013. PMID: 23588220
-
Targeting RPTPσ with lentiviral shRNA promotes neurites outgrowth of cortical neurons and improves functional recovery in a rat spinal cord contusion model.Brain Res. 2014 Oct 24;1586:46-63. doi: 10.1016/j.brainres.2014.08.048. Epub 2014 Aug 21. Brain Res. 2014. PMID: 25152470
-
Regulation of autophagy by inhibitory CSPG interactions with receptor PTPσ and its impact on plasticity and regeneration after spinal cord injury.Exp Neurol. 2020 Jun;328:113276. doi: 10.1016/j.expneurol.2020.113276. Epub 2020 Mar 4. Exp Neurol. 2020. PMID: 32145250 Free PMC article. Review.
-
Molecular mechanisms of scar-sourced axon growth inhibitors.Brain Res. 2015 Sep 4;1619:22-35. doi: 10.1016/j.brainres.2014.08.064. Epub 2014 Sep 1. Brain Res. 2015. PMID: 25192646 Free PMC article. Review.
Cited by
-
Translational Regenerative Therapies for Chronic Spinal Cord Injury.Int J Mol Sci. 2018 Jun 15;19(6):1776. doi: 10.3390/ijms19061776. Int J Mol Sci. 2018. PMID: 29914060 Free PMC article. Review.
-
Modulation of stress-, pain-, and alcohol-related behaviors by perineuronal nets.Neurobiol Stress. 2024 Nov 14;33:100692. doi: 10.1016/j.ynstr.2024.100692. eCollection 2024 Nov. Neurobiol Stress. 2024. PMID: 39691634 Free PMC article.
-
Skeletal muscle TFEB signaling promotes central nervous system function and reduces neuroinflammation during aging and neurodegenerative disease.Cell Rep. 2023 Nov 28;42(11):113436. doi: 10.1016/j.celrep.2023.113436. Epub 2023 Nov 11. Cell Rep. 2023. PMID: 37952157 Free PMC article.
-
Fabrication of homotypic neural ribbons as a multiplex platform optimized for spinal cord delivery.Sci Rep. 2020 Jul 31;10(1):12939. doi: 10.1038/s41598-020-69274-7. Sci Rep. 2020. PMID: 32737387 Free PMC article.
-
Cathepsin B inhibition blocks neurite outgrowth in cultured neurons by regulating lysosomal trafficking and remodeling.J Neurochem. 2020 Nov;155(3):300-312. doi: 10.1111/jnc.15032. Epub 2020 Jun 15. J Neurochem. 2020. PMID: 32330298 Free PMC article.
References
-
- Bijata M, Labus J, Guseva D, Stawarski M, Butzlaff M, Dzwonek J, Schneeberg J, Böhm K, Michaluk P, Rusakov DA, Dityatev A, Wilczyński G, Wlodarczyk J, Ponimaskin E (2017) Synaptic remodeling depends on signaling between serotonin receptors and the extracellular matrix. Cell Rep 19:1767–1782. 10.1016/j.celrep.2017.05.023 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
