Suspected unexpected and other adverse reactions to antiretroviral drugs used as post-exposure prophylaxis of HIV infection - five-year experience from clinical practice
- PMID: 29765441
- PMCID: PMC5949906
- DOI: 10.5114/aoms.2016.59701
Suspected unexpected and other adverse reactions to antiretroviral drugs used as post-exposure prophylaxis of HIV infection - five-year experience from clinical practice
Abstract
Introduction: With increased use of antiretroviral drugs (ARVs) in HIV uninfected persons, proper reporting on suspected unexpected serious adverse reactions (SUSARs) and continued insight into adverse drug reactions (ADRs) are needed for adequate information on safety of ARVs in such populations.
Material and methods: Medical documentation of persons receiving ARVs after non-occupational HIV exposure (non-occupational post-exposure prophylaxis - nPEP) during 5 successive years (2009-2013) was evaluated by two HIV physicians. Adverse drug reactions s and SUSARs were defined according to international standards. In statistical analyses Cox proportional hazard models were used to identify independent predictors of developing a first ADR.
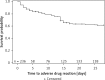
Results: In total 375 persons received nPEP with the following indications: needle stick (43%), unprotected sexual intercourse (17%), rape (10%) and first aid (10%). In 84 (22%) cases the source patient was HIV positive or an active injecting drug user. In total 170 ADRs were reported. One hundred thirty-nine persons had only 1 ADR. The most frequent first ADRs were gastrointestinal disorders (22%), followed by general symptoms (9%), hypersensitivity reactions (1.6%) and CNS symptoms (1.3%). The remaining events represented less than 1% of all patients. Eight (2.1%) patients developed a SUSAR. In multivariate analyses only age at first visit to the clinic was an independent predictor of developing an ADR (HR = 1.17, 95% CI: 1.03-1.34; p = 0.02).
Conclusions: In our observations ADRs in reaction to nPEP were frequent yet usually mild events, mostly occurring in the first 2 weeks and rarely causing discontinuation. The only significant factor increasing the risk of ADR was age. SUSARs were rare, transient and clinically insignificant.
Keywords: HIV prophylaxis; adverse drug reaction; post-exposure prophylaxis; suspected unexpected serious adverse reactions.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Sonder GJ, van den Hoek A, Regez RM, et al. Trends in HIV postexposure prophylaxis prescription and compliance after sexual exposure in Amsterdam, 2000-2004. Sex Transm Dis. 2007;34:288–93. - PubMed
-
- Lunding S, Katzenstein TL, Kronborg G, et al. The Danish PEP registry: experience with the use of postexposure prophylaxis (PEP) following sexual exposure to HIV from 1998 to 2006. Sex Transm Dis. 2010;37:49–52.. - PubMed
-
- Service USPH Updated U.S. Public Health Service Guidelines for the Management of Occupational Exposures to HBV, HCV, and HIV and Recommendations for Postexposure Prophylaxis. MMWR Recomm Rep. 2001;50(RR-11):1–52.. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials