Binding of sulphonylureas to plasma proteins - A KATP channel perspective
- PMID: 29772022
- PMCID: PMC5957440
- DOI: 10.1371/journal.pone.0197634
Binding of sulphonylureas to plasma proteins - A KATP channel perspective
Abstract
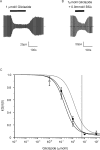
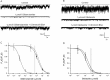
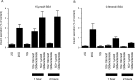
Sulphonylurea drugs stimulate insulin secretion from pancreatic β-cells primarily by inhibiting ATP sensitive potassium (KATP) channels in the β-cell membrane. The effective sulphonylurea concentration at its site of action is significantly attenuated by binding to serum albumin, which makes it difficult to compare in vitro and in vivo data. We therefore measured the ability of gliclazide and glibenclamide to inhibit KATP channels and stimulate insulin secretion in the presence of serum albumin. We used this data, together with estimates of free drug concentrations from binding studies, to predict the extent of sulphonylurea inhibition of KATP channels at therapeutic concentrations in vivo. KATP currents from mouse pancreatic β-cells and Xenopus oocytes were measured using the patch-clamp technique. Gliclazide and glibenclamide binding to human plasma were determined in spiked plasma samples using an ultrafiltration-mass spectrometry approach. Bovine serum albumin (60g/l) produced a mild, non-significant reduction of gliclazide block of KATP currents in pancreatic β-cells and Xenopus oocytes. In contrast, glibenclamide inhibition of recombinant KATP channels was dramatically suppressed by albumin (predicted free drug concentration <0.1%). Insulin secretion was also reduced. Free concentrations of gliclazide and glibenclamide in the presence of human plasma measured in binding experiments were 15% and 0.05%, respectively. Our data suggest the free concentration of glibenclamide in plasma is too low to account for the drug's therapeutic effect. In contrast, the free gliclazide concentration in plasma is high enough to close KATP channels and stimulate insulin secretion.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Molecular mechanism of sulphonylurea block of K(ATP) channels carrying mutations that impair ATP inhibition and cause neonatal diabetes.Diabetes. 2013 Nov;62(11):3909-19. doi: 10.2337/db13-0531. Epub 2013 Jul 8. Diabetes. 2013. PMID: 23835339 Free PMC article.
-
Taurine block of cloned ATP-sensitive K+ channels with different sulfonylurea receptor subunits expressed in Xenopus laevis oocytes.Biochem Pharmacol. 2004 Sep 1;68(5):901-10. doi: 10.1016/j.bcp.2004.05.050. Biochem Pharmacol. 2004. PMID: 15294453
-
Gliclazide produces high-affinity block of KATP channels in mouse isolated pancreatic beta cells but not rat heart or arterial smooth muscle cells.Diabetologia. 2001 Aug;44(8):1019-25. doi: 10.1007/s001250100595. Diabetologia. 2001. PMID: 11484080
-
Role of K(ATP) channels in β-cell resistance to oxidative stress.Diabetes Obes Metab. 2012 Oct;14 Suppl 3:120-8. doi: 10.1111/j.1463-1326.2012.01644.x. Diabetes Obes Metab. 2012. PMID: 22928572 Review.
-
Characterization of the molecular mode of action of the sulfonylurea, glimepiride, at beta-cells.Horm Metab Res. 1996 Sep;28(9):464-8. doi: 10.1055/s-2007-979838. Horm Metab Res. 1996. PMID: 8911984 Review.
Cited by
-
Combining Sulfonylureas with Anticancer Drugs: Evidence of Synergistic Efficacy with Doxorubicin In Vitro and In Vivo.Int J Mol Sci. 2025 Feb 8;26(4):1429. doi: 10.3390/ijms26041429. Int J Mol Sci. 2025. PMID: 40003896 Free PMC article.
-
Computational studies for pre-evaluation of pharmacological profile of gut microbiota-produced gliclazide metabolites.Front Pharmacol. 2024 Dec 3;15:1492284. doi: 10.3389/fphar.2024.1492284. eCollection 2024. Front Pharmacol. 2024. PMID: 39691391 Free PMC article.
-
Vitamin D Supplementation Could Enhance the Effectiveness of Glibenclamide in Treating Diabetes and Preventing Diabetic Nephropathy: A Biochemical, Histological and Immunohistochemical Study.J Evid Based Integr Med. 2022 Jan-Dec;27:2515690X221116403. doi: 10.1177/2515690X221116403. J Evid Based Integr Med. 2022. PMID: 35942573 Free PMC article.
-
A systematic analysis of anti-diabetic medicinal plants from cells to clinical trials.PeerJ. 2023 Jan 5;11:e14639. doi: 10.7717/peerj.14639. eCollection 2023. PeerJ. 2023. PMID: 36627919 Free PMC article. Review.
-
Combination Therapy of Alpha-Lipoic Acid, Gliclazide and Ramipril Protects Against Development of Diabetic Cardiomyopathy via Inhibition of TGF-β/Smad Pathway.Front Pharmacol. 2022 Mar 21;13:850542. doi: 10.3389/fphar.2022.850542. eCollection 2022. Front Pharmacol. 2022. PMID: 35401218 Free PMC article.
References
-
- Zünkler BJ, Lenzen S, Männer K, Panten U, Trube G. Concentration-dependent effects of tolbutamide, meglitinide, glipizide, glibenclamide and diazoxide on ATP-regulated K+ currents in pancreatic B-cells. Naunyn Schmiedebergs Arch Pharmacol. 1988;337: 225–230. - PubMed
-
- Gribble FM, Reimann F. Sulphonylurea action revisited: the post-cloning era. Diabetologia. 2003;46: 875–891. doi: 10.1007/s00125-003-1143-3 - DOI - PubMed
-
- Martin GM, Kandasamy B, DiMaio F, Yoshioka C, Shyng S. Anti-diabetic drug binding site in a mammalian KATP channel revealed by Cryo-EM. Elife. 2017; 6: e31054 doi: 10.7554/eLife.31054 - DOI - PMC - PubMed
-
- Ashcroft FM, Rorsman P. Electrophysiology of the pancreatic beta cell. Prog Biophys Mol Biol. 1989;54: 87–143. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

