Influence of blood flow on cardiac development
- PMID: 29772208
- PMCID: PMC6109420
- DOI: 10.1016/j.pbiomolbio.2018.05.005
Influence of blood flow on cardiac development
Abstract
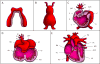
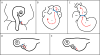
The role of hemodynamics in cardiovascular development is not well understood. Indeed, it would be remarkable if it were, given the dauntingly complex array of intricately synchronized genetic, molecular, mechanical, and environmental factors at play. However, with congenital heart defects affecting around 1 in 100 human births, and numerous studies pointing to hemodynamics as a factor in cardiovascular morphogenesis, this is not an area in which we can afford to remain in the dark. This review seeks to present the case for the importance of research into the biomechanics of the developing cardiovascular system. This is accomplished by i) illustrating the basics of some of the highly complex processes involved in heart development, and discussing the known influence of hemodynamics on those processes; ii) demonstrating how altered hemodynamic environments have the potential to bring about morphological anomalies, citing studies in multiple animal models with a variety of perturbation methods; iii) providing examples of widely used technological innovations which allow for accurate measurement of hemodynamic parameters in embryos; iv) detailing the results of studies in avian embryos which point to exciting correlations between various hemodynamic manipulations in early development and phenotypic defect incidence in mature hearts; and finally, v) stressing the relevance of uncovering specific biomechanical pathways involved in cardiovascular formation and remodeling under adverse conditions, to the potential treatment of human patients. The time is ripe to unravel the contributions of hemodynamics to cardiac development, and to recognize their frequently neglected role in the occurrence of heart malformation phenotypes.
Keywords: Congenital heart disease; Heart formation; Hemodynamics; Mechanotransduction.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Figures




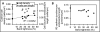

References
-
- [Accessed November 20, 2017];Measure hydrostatic pressure in small vessels and oocytes. [Online]. https://www.wpiinc.com/products/top-products/sys-900a-micropressure-system/
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

