Survival prediction based on qualitative MRI diffusion signature in patients with recurrent high grade glioma treated with bevacizumab
- PMID: 29774180
- PMCID: PMC5941208
- DOI: 10.21037/qims.2018.04.05
Survival prediction based on qualitative MRI diffusion signature in patients with recurrent high grade glioma treated with bevacizumab
Abstract
Background: Bevacizumab was approved by the FDA for the treatment of recurrent or progressive glioblastoma (GBM). Imaging responses are typically assessed by gadolinium-enhanced MRI. We sought to determine the significance of qualitative diffusion signature (manifest as variable degree of dark signal) on ADC maps in recurrent gliomas after treatment with bevacizumab.
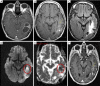
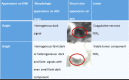
Methods: We performed an institutional review board (IRB) approved retrospective study on patients who underwent MRI of the brain after 8 weeks of receiving bevacizumab for recurrent glioma. Patients were divided into three groups based on qualitative diffusion signature: (I) lesion not bright on diffusion weighted imaging (DWI) suggestive of no restricted diffusion (FDR0); (II) lesion bright on DWI with corresponding homogenous dark signal on apparent diffusion coefficient (ADC) maps suggestive of focal restricted diffusion likely due to bevacizumab induced necrosis (FDRn); and (III) lesion bright on DWI with corresponding homogenous faint dark signal on ADC maps suggestive of focal restricted diffusion likely due to viable tumor or heterogeneous spectrum of dark and faint dark signals on ADC maps suggestive of focal restricted diffusion likely due to viable tumor surrounding the bevacizumab induced necrosis (FDRt).
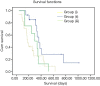
Results: Based on the qualitative signal on diffusion weighted sequences after bevacizumab therapy, total number of patients in group (I) were 14 (36%), in group (II) were 17 (44%); and in group (III) were 8 (20%). The median overall survival (OS) from the time of recurrence in patients belonging to group (II) was 364 days vs. 183 days for those with group (I) vs. 298 days for group (III). On simultaneous comparison of survival differences in all three groups by Kaplan-Meier analysis, group (II) was significant in predicting survival with P values for the log-rank tests <0.033.
Conclusions: In patients with recurrent glioma treated with bevacizumab, the presence of homogenous dark signal (FDRn) on ADC maps at 8 weeks follow-up MRI correlated with a longer survival. Thus, use of this qualitative diffusion signature in adjunct to contrast enhanced MRI may have the widest potential impact on routine clinical care for patients with recurrent high-grade gliomas. However, prospective studies analysing its predictive value are warranted.
Keywords: Recurrent glioblastoma multiforme (GBM); apparent diffusion coefficient (ADC); bevacizumab; diffusion weighted imaging (DWI); median survival.
Conflict of interest statement
Conflicts of Interest: The authors have no conflicts of interest to declare.
Figures

Similar articles
-
The Use of Apparent Diffusion Coefficient Values for Differentiating Bevacizumab-Related Cytotoxicity from Tumor Recurrence and Radiation Necrosis in Glioblastoma.Cancers (Basel). 2024 Jul 2;16(13):2440. doi: 10.3390/cancers16132440. Cancers (Basel). 2024. PMID: 39001500 Free PMC article.
-
Graded functional diffusion map-defined characteristics of apparent diffusion coefficients predict overall survival in recurrent glioblastoma treated with bevacizumab.Neuro Oncol. 2011 Oct;13(10):1151-61. doi: 10.1093/neuonc/nor079. Epub 2011 Aug 19. Neuro Oncol. 2011. PMID: 21856685 Free PMC article.
-
Apparent diffusion coefficient and tumor volume measurements help stratify progression-free survival of bevacizumab-treated patients with recurrent glioblastoma multiforme.Neuroradiol J. 2019 Aug;32(4):241-249. doi: 10.1177/1971400919847184. Epub 2019 May 8. Neuroradiol J. 2019. PMID: 31066622 Free PMC article.
-
Diagnostic Effect of MRI Diffusion-weighted Imaging Apparent Diffusion Coefficient Value on Postoperative Recurrence of Brain Glioma.Altern Ther Health Med. 2023 Oct 27:AT9342. Online ahead of print. Altern Ther Health Med. 2023. PMID: 37883754
-
Imaging biomarkers guided anti-angiogenic therapy for malignant gliomas.Neuroimage Clin. 2018 Jul 5;20:51-60. doi: 10.1016/j.nicl.2018.07.001. eCollection 2018. Neuroimage Clin. 2018. PMID: 30069427 Free PMC article. Review.
Cited by
-
A novel DNA repair-related nomogram predicts survival in low-grade gliomas.CNS Neurosci Ther. 2021 Feb;27(2):186-195. doi: 10.1111/cns.13464. Epub 2020 Oct 16. CNS Neurosci Ther. 2021. PMID: 33063446 Free PMC article.
-
The MRI Features and Prognosis of Gliomas Associated With IDH1 Mutation: A Single Center Study in Southwest China.Front Oncol. 2020 Jun 2;10:852. doi: 10.3389/fonc.2020.00852. eCollection 2020. Front Oncol. 2020. PMID: 32582544 Free PMC article.
-
Early assessment of recurrent glioblastoma response to bevacizumab treatment by diffusional kurtosis imaging: a preliminary report.Neuroradiol J. 2019 Oct;32(5):317-327. doi: 10.1177/1971400919861409. Epub 2019 Jul 8. Neuroradiol J. 2019. PMID: 31282311 Free PMC article.
-
Bevacizumab dose adjustment to improve clinical outcomes of glioblastoma.BMC Med. 2020 Jun 22;18(1):142. doi: 10.1186/s12916-020-01610-0. BMC Med. 2020. PMID: 32564774 Free PMC article.
-
Reactive oxygen species metabolism-based prediction model and drug for patients with recurrent glioblastoma.Aging (Albany NY). 2019 Dec 4;11(23):11010-11029. doi: 10.18632/aging.102506. Epub 2019 Dec 4. Aging (Albany NY). 2019. PMID: 31801111 Free PMC article.
References
-
- Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups ;National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005;352:987-996. 10.1056/NEJMoa043330 - DOI - PubMed
-
- Lamborn KR, Yung WK, Chang SM, Wen PY, Cloughesy TF, DeAngelis LM, Robins HI, Lieberman FS, Fine HA, Fink KL, Junck L, Abrey L, Gilbert MR, Mehta M, Kuhn JG, Aldape KD, Hibberts J, Peterson PM, Prados MD, North American Brain Tumor Consortium Progression-free survival: an important end point in evaluating therapy for recurrent high-grade gliomas. Neuro-oncology 2008;10:162-70. 10.1215/15228517-2007-062 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
