Temporary microglia-depletion after cosmic radiation modifies phagocytic activity and prevents cognitive deficits
- PMID: 29777152
- PMCID: PMC5959907
- DOI: 10.1038/s41598-018-26039-7
Temporary microglia-depletion after cosmic radiation modifies phagocytic activity and prevents cognitive deficits
Erratum in
-
Author Correction: Temporary microglia-depletion after cosmic radiation modifies phagocytic activity and prevents cognitive deficits.Sci Rep. 2018 Jul 3;8(1):10297. doi: 10.1038/s41598-018-28390-1. Sci Rep. 2018. PMID: 29967490 Free PMC article.
Abstract
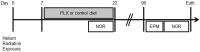
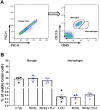
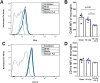
Microglia are the main immune component in the brain that can regulate neuronal health and synapse function. Exposure to cosmic radiation can cause long-term cognitive impairments in rodent models thereby presenting potential obstacles for astronauts engaged in deep space travel. The mechanism/s for how cosmic radiation induces cognitive deficits are currently unknown. We find that temporary microglia depletion, one week after cosmic radiation, prevents the development of long-term memory deficits. Gene array profiling reveals that acute microglia depletion alters the late neuroinflammatory response to cosmic radiation. The repopulated microglia present a modified functional phenotype with reduced expression of scavenger receptors, lysosome membrane protein and complement receptor, all shown to be involved in microglia-synapses interaction. The lower phagocytic activity observed in the repopulated microglia is paralleled by improved synaptic protein expression. Our data provide mechanistic evidence for the role of microglia in the development of cognitive deficits after cosmic radiation exposure.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

