Spontaneous Hair Cell Regeneration Is Prevented by Increased Notch Signaling in Supporting Cells
- PMID: 29780306
- PMCID: PMC5945818
- DOI: 10.3389/fncel.2018.00120
Spontaneous Hair Cell Regeneration Is Prevented by Increased Notch Signaling in Supporting Cells
Abstract
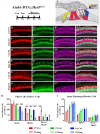
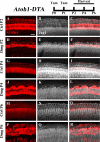
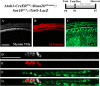
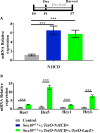
During embryonic development, differentiation of cochlear progenitor cells into hair cells (HCs) or supporting cells (SCs) is partially controlled through Notch signaling. Many studies have shown that inhibition of Notch signaling allows SCs to convert into HCs in both normal and drug damaged neonatal mouse cochleae. This mechanism is also implicated during HC regeneration in non-mammalian vertebrates; however, the mechanism of spontaneous HC regeneration in the neonatal mouse cochlea is less understood. While inhibition of Notch signaling can force SCs to convert into HCs and increase the number of regenerated HCs, it is currently unknown whether this pathway is involved in spontaneous HC regeneration observed in vivo. Therefore, we investigated the role of Notch signaling during the spontaneous HC regeneration process using Atoh1-CreERTM::Rosa26loxP-stop-loxP-DTA/+ mice injected with tamoxifen at postnatal day (P) 0 and P1 to ablate HCs and stimulate spontaneous HC regeneration. Expression changes of genes in the Notch pathway were measured using immunostaining and in situ hybridization, with most changes observed in the apical one-third of the cochlea where the majority of HC regeneration occurs. Expression of the Notch target genes Hes1, Hes5, Hey1, HeyL, and Jagged1 were decreased. To investigate whether reduction of Notch signaling is involved in the spontaneous HC regeneration process, we overexpressed the Notch1 intracellular fragment (N1ICD) in cochlear SCs and other non-sensory epithelial cells in the context of HC damage. Specifically, Atoh1-CreERTM::Rosa26loxP-stop-loxP-DTA/+::Sox10rtTA::TetO-LacZ::TetO-N1ICD mice were injected with tamoxifen at P0/P1 to stimulate spontaneous HC regeneration and given doxycycline from P0-P7 to induce expression of N1ICD as well as LacZ for fate-mapping. We observed a 92% reduction in the number of fate-mapped regenerated HCs in mice with N1ICD overexpression compared to controls with HC damage but no manipulation of Notch signaling. Therefore, we conclude that increased Notch signaling prevents spontaneous HC regeneration from occurring in the neonatal mouse cochlea. Understanding which components of the Notch pathway regulates regenerative plasticity in the neonatal mouse cochlea will inform investigations focused on stimulating HC regeneration in mature cochlea and eventually in humans to treat hearing loss.
Keywords: CreER; NICD; Notch; Tet-On; cochlea; hair cell regeneration.
Figures


Similar articles
-
Overactivation of Notch1 signaling induces ectopic hair cells in the mouse inner ear in an age-dependent manner.PLoS One. 2012;7(3):e34123. doi: 10.1371/journal.pone.0034123. Epub 2012 Mar 20. PLoS One. 2012. PMID: 22448289 Free PMC article.
-
Conditional overexpression of neuritin in supporting cells (SCs) mitigates hair cell (HC) damage and induces HC regeneration in the adult mouse cochlea after drug-induced ototoxicity.Hear Res. 2022 Jul;420:108515. doi: 10.1016/j.heares.2022.108515. Epub 2022 May 11. Hear Res. 2022. PMID: 35584572
-
Extensive Supporting Cell Proliferation and Mitotic Hair Cell Generation by In Vivo Genetic Reprogramming in the Neonatal Mouse Cochlea.J Neurosci. 2016 Aug 17;36(33):8734-45. doi: 10.1523/JNEUROSCI.0060-16.2016. J Neurosci. 2016. PMID: 27535918 Free PMC article.
-
Advances in Understanding the Notch Signaling Pathway in the Cochlea.Curr Pharm Des. 2023;29(41):3266-3273. doi: 10.2174/0113816128273532231103110910. Curr Pharm Des. 2023. PMID: 37990430 Review.
-
Therapeutic Potential of Wnt and Notch Signaling and Epigenetic Regulation in Mammalian Sensory Hair Cell Regeneration.Mol Ther. 2019 May 8;27(5):904-911. doi: 10.1016/j.ymthe.2019.03.017. Epub 2019 Mar 30. Mol Ther. 2019. PMID: 30982678 Free PMC article. Review.
Cited by
-
Kolliker's Organ and Its Functional Role in the Development of Corti's Organ and Auditory Systems.Audiol Res. 2025 Jun 23;15(4):75. doi: 10.3390/audiolres15040075. Audiol Res. 2025. PMID: 40700218 Free PMC article. Review.
-
Approaches for the study of epigenetic modifications in the inner ear and related tissues.Hear Res. 2019 May;376:69-85. doi: 10.1016/j.heares.2019.01.007. Epub 2019 Jan 12. Hear Res. 2019. PMID: 30679030 Free PMC article. Review.
-
Sponge bHLH Gene Expression in Xenopus laevis Disrupts Inner Ear and Lateral Line Neurosensory Development and Otic Afferent Pathfinding.Int J Mol Sci. 2025 Jun 7;26(12):5487. doi: 10.3390/ijms26125487. Int J Mol Sci. 2025. PMID: 40564948 Free PMC article.
-
Uncovering Novel Prognostic Factors of Sudden Sensorineural Hearing Loss by Whole-Genome Sequencing of Cell-Free DNA.J Int Adv Otol. 2022 Nov;18(6):459-464. doi: 10.5152/iao.2022.21493. J Int Adv Otol. 2022. PMID: 36349665 Free PMC article.
-
Characterizing Adult Cochlear Supporting Cell Transcriptional Diversity Using Single-Cell RNA-Seq: Validation in the Adult Mouse and Translational Implications for the Adult Human Cochlea.Front Mol Neurosci. 2020 Feb 5;13:13. doi: 10.3389/fnmol.2020.00013. eCollection 2020. Front Mol Neurosci. 2020. PMID: 32116546 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

