Extracellular vesicles extracted from young donor serum attenuate inflammaging via partially rejuvenating aged T-cell immunotolerance
- PMID: 29782203
- PMCID: PMC6181631
- DOI: 10.1096/fj.201800059R
Extracellular vesicles extracted from young donor serum attenuate inflammaging via partially rejuvenating aged T-cell immunotolerance
Abstract
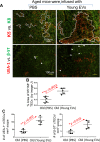
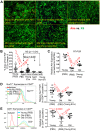
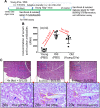
Biologic aging results in a chronic inflammatory condition, termed inflammaging, which establishes a risk for such age-related diseases as neurocardiovascular diseases; therefore, it is of great importance to develop rejuvenation strategies that are able to attenuate inflammaging as a means of intervention for age-related diseases. A promising rejuvenation factor that is present in young blood has been found that can make aged neurons younger; however, the component in the young blood and its mechanism of action are poorly elucidated. We assessed rejuvenation in naturally aged mice with extracellular vesicles (EVs) or exosomes extracted from young murine serum on the basis of different spectrums of microRNAs in these vesicles from young and old sera. We found that EVs extracted from young donor mouse serum, rather than EVs extracted from old donor mouse serum or non-EV supernatant extracted from young donor mouse serum, were able to attenuate inflammaging in old mice. Inflammaging is attributed to multiple factors, one of which is thymic aging-released self-reactive T cell-induced pathology. We found that the attenuation of inflammaging after treatment with EVs from young serum partially contributed to the rejuvenation of thymic aging, which is characterized by partially reversed thymic involution, enhancement of negative selection signals, and reduced autoreactions in the periphery. Our results provide evidence for understanding of the potential rejuvenation factor in the young donor serum, which holds great promise for the development of novel therapeutics to reduce morbidity and mortality caused by age-related inflammatory diseases.-Wang, W., Wang, L., Ruan, L., Oh, J., Dong, X., Zhuge, Q., Su, D.-M. Extracellular vesicles extracted from young donor serum attenuate inflammaging via partially rejuvenating aged T-cell immunotolerance.
Keywords: age-related thymic involution; chronic inflammation; exosomes; rejuvenation; young serum.
Conflict of interest statement
This work was supported, in part, by U.S. National Institutes of Health (NIH), National Institute of Allergy and Infectious Diseases Grant R01-AI121147 (to D.-M.S.), and by the National Natural Science Foundation of China (31660256; to L.W.). The authors declare no conflicts of interest.
W. Wang performed most of the experiments and prepared figures and the manuscript; L. Wang and L. Ruan performed part of experiments related to inflammation; J. Oh performed part of experiments related to gene expression; X. Dong performed EV and exosome analysis; Q. Zhuge provided instruction on studies of neuronal inflammation; and D.-M. Su designed the overall research project, instructed experiments, data analysis, and wrote the manuscript.
Figures
Similar articles
-
Contributions of Age-Related Thymic Involution to Immunosenescence and Inflammaging.Immun Ageing. 2020 Jan 20;17:2. doi: 10.1186/s12979-020-0173-8. eCollection 2020. Immun Ageing. 2020. PMID: 31988649 Free PMC article. Review.
-
Thymic involution perturbs negative selection leading to autoreactive T cells that induce chronic inflammation.J Immunol. 2015 Jun 15;194(12):5825-37. doi: 10.4049/jimmunol.1500082. Epub 2015 May 8. J Immunol. 2015. PMID: 25957168 Free PMC article.
-
Preconditioned Mesenchymal Stromal Cell-Derived Extracellular Vesicles (EVs) Counteract Inflammaging.Cells. 2022 Nov 21;11(22):3695. doi: 10.3390/cells11223695. Cells. 2022. PMID: 36429124 Free PMC article.
-
The profile of circulating extracellular vesicles depending on the age of the donor potentially drives the rejuvenation or senescence fate of hematopoietic stem cells.Exp Gerontol. 2023 May;175:112142. doi: 10.1016/j.exger.2023.112142. Epub 2023 Mar 22. Exp Gerontol. 2023. PMID: 36921675
-
Senescent cell-derived extracellular vesicles as potential mediators of innate immunosenescence and inflammaging.Exp Gerontol. 2024 Mar;187:112365. doi: 10.1016/j.exger.2024.112365. Epub 2024 Jan 24. Exp Gerontol. 2024. PMID: 38237747 Review.
Cited by
-
Thymic atrophy creates holes in Treg-mediated immuno-regulation via impairment of an antigen-specific clone.Immunology. 2021 Aug;163(4):478-492. doi: 10.1111/imm.13333. Epub 2021 Apr 15. Immunology. 2021. PMID: 33786850 Free PMC article.
-
From Genesis to Old Age: Exploring the Immune System One Cell at a Time with Flow Cytometry.Biomedicines. 2024 Jul 3;12(7):1469. doi: 10.3390/biomedicines12071469. Biomedicines. 2024. PMID: 39062042 Free PMC article. Review.
-
Thymic Aging May Be Associated with COVID-19 Pathophysiology in the Elderly.Cells. 2021 Mar 12;10(3):628. doi: 10.3390/cells10030628. Cells. 2021. PMID: 33808998 Free PMC article. Review.
-
Cellular expansion of MSCs: Shifting the regenerative potential.Aging Cell. 2023 Jan;22(1):e13759. doi: 10.1111/acel.13759. Epub 2022 Dec 19. Aging Cell. 2023. PMID: 36536521 Free PMC article. Review.
-
MicroRNA Functions in Thymic Biology: Thymic Development and Involution.Front Immunol. 2018 Sep 11;9:2063. doi: 10.3389/fimmu.2018.02063. eCollection 2018. Front Immunol. 2018. PMID: 30254640 Free PMC article. Review.
References
-
- Villeda S. A., Plambeck K. E., Middeldorp J., Castellano J. M., Mosher K. I., Luo J., Smith L. K., Bieri G., Lin K., Berdnik D., Wabl R., Udeochu J., Wheatley E. G., Zou B., Simmons D. A., Xie X. S., Longo F. M., Wyss-Coray T. (2014) Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice. Nat. Med. 20, 659–663 - PMC - PubMed
-
- Conboy I. M., Conboy M. J., Wagers A. J., Girma E. R., Weissman I. L., Rando T. A. (2005) Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature 433, 760–764 - PubMed
-
- Pishel I., Shytikov D., Orlova T., Peregudov A., Artyuhov I., Butenko G. (2012) Accelerated aging versus rejuvenation of the immune system in heterochronic parabiosis. Rejuvenation Res. 15, 239–248 - PubMed
-
- Brack A. S., Conboy M. J., Roy S., Lee M., Kuo C. J., Keller C., Rando T. A. (2007) Increased Wnt signaling during aging alters muscle stem cell fate and increases fibrosis. Science 317, 807–810 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

