Induction of multiple myeloma cancer stem cell apoptosis using conjugated anti-ABCG2 antibody with epirubicin-loaded microbubbles
- PMID: 29784015
- PMCID: PMC5963075
- DOI: 10.1186/s13287-018-0885-2
Induction of multiple myeloma cancer stem cell apoptosis using conjugated anti-ABCG2 antibody with epirubicin-loaded microbubbles
Abstract
Background: Multiple myeloma (MM) currently remains largely incurable. Cancer stem cells (CSCs) are believed to be responsible for drug resistance and eventual relapse. In this study, we exploited a novel agent to evaluate its inhibitory effect on MM CSCs.
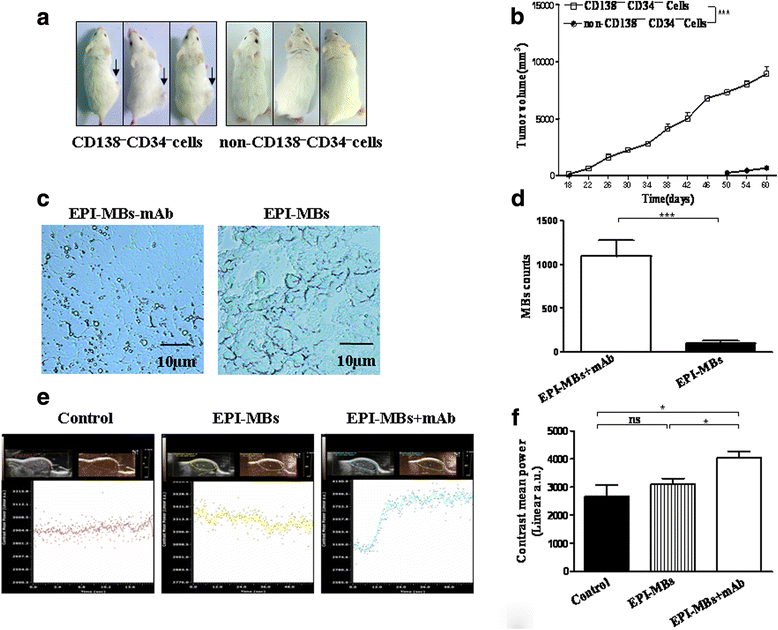
Methods: Epirubicin (EPI)-loaded lipid microbubbles (MBs) conjugated with anti-ABCG2 monoclonal antibody (EPI-MBs + mAb) were developed and their effect on MM 138-CD34- CSCs isolated from human MM RPMI 8226 cell line plus ultrasound exposure in vitro and in vivo in a nonobese diabetic/severe combined immunodeficient mouse model were assessed.
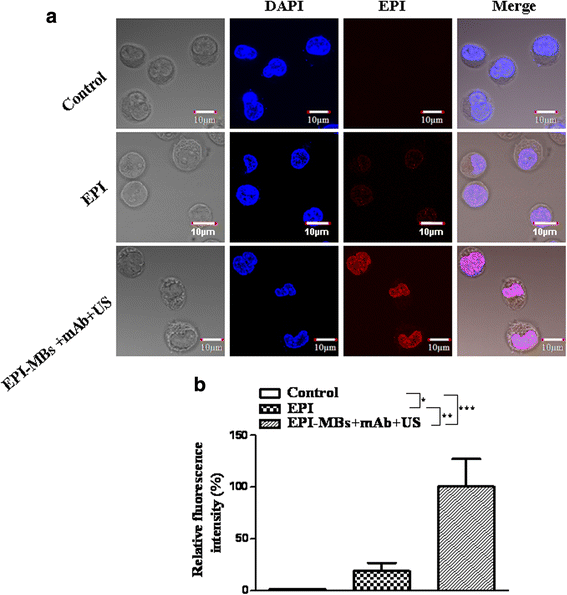
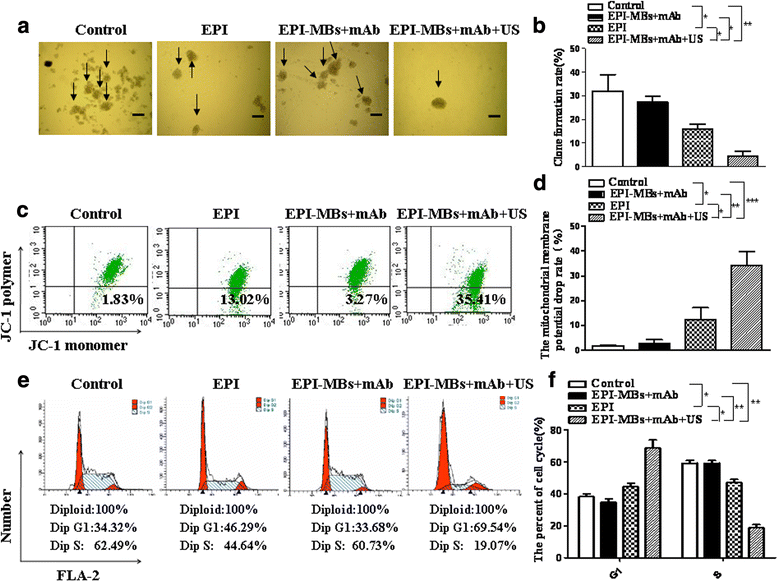
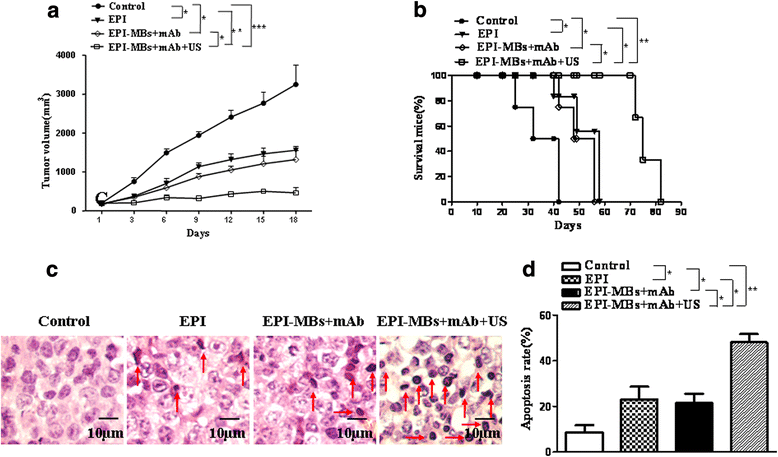
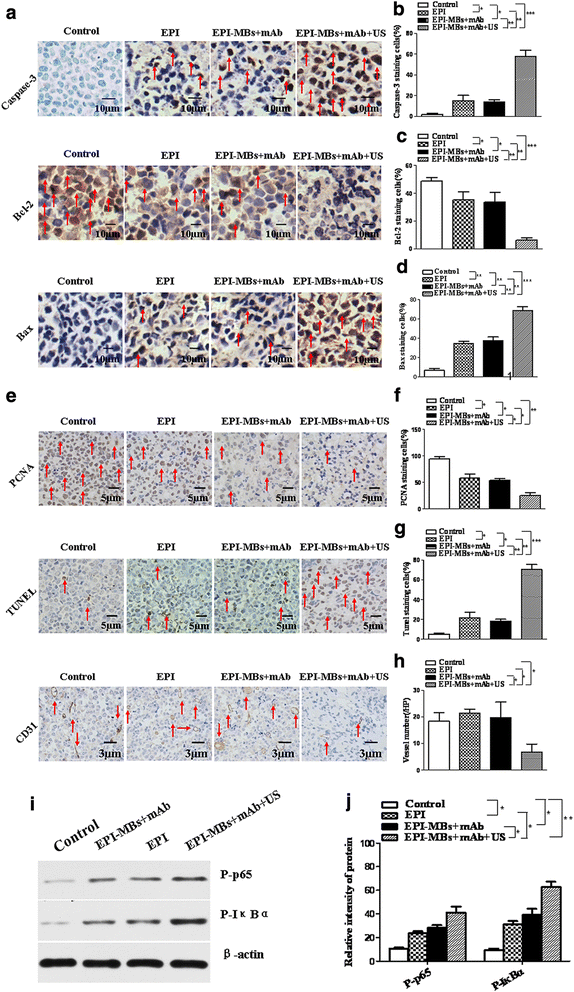
Results: EPI-MBs + mAb combined with ultrasound led to a significant decrease in the clone formation ability and the mitochondrial membrane potential along with an increase in MM CSC apoptosis. Moreover, treatment with EPI-MBs + mAb with ultrasound exposure remarkably inhibited the growth of MM CSC-derived tumors in xenograft nonobese diabetic/severe combined immunodeficient mice compared with a single agent or EPI-MBs + mAb without ultrasound exposure. The inhibitive efficacy was also correlated with an increased expression of caspase-3, Bax, and TUNEL and decreased expressions of PCNA, Bcl-2, and CD31.
Conclusions: Our findings reveal that the EPI-MBs + mAb combined with therapeutic ultrasound may confer an effective approach for treatment of MM by induction of an apoptotic pathway in MM CSCs.
Keywords: Cancer stem cells; Epirubicin; Microbubble; Multiple myeloma; Ultrasound.
Conflict of interest statement
Ethics approval
Animal experiments were conducted in accordance with the guidelines of the Animal Research Ethics Board of Southeast University. This ethics board also approved the animal studies.
Consent for publication
All authors have contributed to, read, and approved the final manuscript for submission.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Inhibitory effect of epirubicin-loaded lipid microbubbles with conjugated anti-ABCG2 antibody combined with therapeutic ultrasound on multiple myeloma cancer stem cells.J Drug Target. 2016;24(1):34-46. doi: 10.3109/1061186X.2015.1052075. Epub 2015 Jul 23. J Drug Target. 2016. PMID: 26204324
-
Enhancing the anti-multiple myeloma efficiency in a cancer stem cell xenograft model by conjugating the ABCG2 antibody with microbubbles for a targeted delivery of ultrasound mediated epirubicin.Biochem Pharmacol. 2017 May 15;132:18-28. doi: 10.1016/j.bcp.2017.02.014. Epub 2017 Feb 20. Biochem Pharmacol. 2017. PMID: 28232025
-
Gamma-Fe2O3 nanoparticles increase therapeutic efficacy of combination with paclitaxel and anti-ABCG2 monoclonal antibody on multiple myeloma cancer stem cells in mouse model.J Biomed Nanotechnol. 2014 Feb;10(2):336-44. doi: 10.1166/jbn.2014.1730. J Biomed Nanotechnol. 2014. PMID: 24738341
-
Concise review: Defining and targeting myeloma stem cell-like cells.Stem Cells. 2014 May;32(5):1067-73. doi: 10.1002/stem.1643. Stem Cells. 2014. PMID: 24449391 Review.
-
Epirubicin: Biological Properties, Analytical Methods, and Drug Delivery Nanosystems.Crit Rev Anal Chem. 2023;53(5):1080-1093. doi: 10.1080/10408347.2021.2007469. Epub 2021 Nov 24. Crit Rev Anal Chem. 2023. PMID: 34818953 Review.
Cited by
-
Iron Oxide Nanoparticles Combined with Cytosine Arabinoside Show Anti-Leukemia Stem Cell Effects on Acute Myeloid Leukemia by Regulating Reactive Oxygen Species.Int J Nanomedicine. 2021 Feb 17;16:1231-1244. doi: 10.2147/IJN.S278885. eCollection 2021. Int J Nanomedicine. 2021. PMID: 33633448 Free PMC article.
-
Plasmonic photothermal release of docetaxel by gold nanoparticles incorporated onto halloysite nanotubes with conjugated 2D8-E3 antibodies for selective cancer therapy.J Nanobiotechnology. 2021 Aug 11;19(1):239. doi: 10.1186/s12951-021-00982-6. J Nanobiotechnology. 2021. PMID: 34380469 Free PMC article.
-
Identification and Characterization of Multiple Myeloma Stem Cell-Like Cells.Cancers (Basel). 2021 Jul 14;13(14):3523. doi: 10.3390/cancers13143523. Cancers (Basel). 2021. PMID: 34298738 Free PMC article. Review.
-
Cyclopamine sensitizes multiple myeloma cells to circularly permuted TRAIL-induced apoptosis.Oncol Lett. 2021 Apr;21(4):295. doi: 10.3892/ol.2021.12556. Epub 2021 Feb 17. Oncol Lett. 2021. PMID: 33732371 Free PMC article.
-
Cancer stem cells and strategies for targeted drug delivery.Drug Deliv Transl Res. 2021 Oct;11(5):1779-1805. doi: 10.1007/s13346-020-00863-9. Epub 2020 Oct 23. Drug Deliv Transl Res. 2021. PMID: 33095384 Free PMC article. Review.
References
-
- Dou J, Li Y, Zhao F, Hu W, Wen P, Tang Q, et al. Identification of tumor stem-like cells in a mouse myeloma cell line. Cell Mol Biol (Noisy-le-grand) 2009;55(Suppl):L1151–L1160. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

