Magnesium Handling in the Kidney
- PMID: 29793662
- PMCID: PMC6932629
- DOI: 10.1053/j.ackd.2018.01.003
Magnesium Handling in the Kidney
Abstract
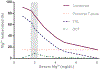
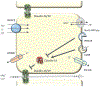
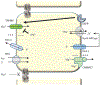
Magnesium is a divalent cation that fills essential roles as regulator and cofactor in a variety of biological pathways, and maintenance of magnesium balance is vital to human health. The kidney, in concert with the intestine, has an important role in maintaining magnesium homeostasis. Although micropuncture and microperfusion studies in the mammalian nephron have shone a light on magnesium handling in the various nephron segments, much of what we know about the protein mediators of magnesium handling in the kidney have come from more recent genetic studies. In the proximal tubule and thick ascending limb, magnesium reabsorption is believed to occur primarily through the paracellular shunt pathway, which ultimately depends on the electrochemical gradient setup by active sodium reabsorption. In the distal convoluted tubule, magnesium transport is transcellular, although magnesium reabsorption also appears to be related to active sodium reabsorption in this segment. In addition, evidence suggests that magnesium transport is highly regulated, although a specific hormonal regulator of extracellular magnesium has yet to be identified.
Keywords: Claudins; Homeostasis; Kidney reabsorption; Magnesium; Nephrons.
Copyright © 2018 National Kidney Foundation, Inc. Published by Elsevier Inc. All rights reserved.
Figures
References
-
- Garfinkel L, Garfinkel D. Calculation of free-Mg2+ concentration in adenosine 5'-triphosphate containing solutions in vitro and in vivo. Biochemistry. 1984;23(15):3547–52. PubMed PMID: 6331847. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

