Control of Heterologous Simian Immunodeficiency Virus SIVsmE660 Infection by DNA and Protein Coimmunization Regimens Combined with Different Toll-Like-Receptor-4-Based Adjuvants in Macaques
- PMID: 29793957
- PMCID: PMC6052320
- DOI: 10.1128/JVI.00281-18
Control of Heterologous Simian Immunodeficiency Virus SIVsmE660 Infection by DNA and Protein Coimmunization Regimens Combined with Different Toll-Like-Receptor-4-Based Adjuvants in Macaques
Abstract
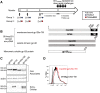
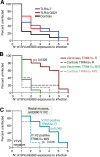
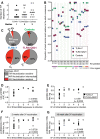
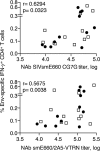
We developed a method of simultaneous vaccination with DNA and protein resulting in robust and durable cellular and humoral immune responses with efficient dissemination to mucosal sites and protection against simian immunodeficiency virus (SIV) infection. To further optimize the DNA-protein coimmunization regimen, we tested a SIVmac251-based vaccine formulated with either of two Toll-like receptor 4 (TLR4) ligand-based liposomal adjuvant formulations (TLR4 plus TLR7 [TLR4+7] or TLR4 plus QS21 [TLR4+QS21]) in macaques. Although both vaccines induced humoral responses of similar magnitudes, they differed in their functional quality, including broader neutralizing activity and effector functions in the TLR4+7 group. Upon repeated heterologous SIVsmE660 challenge, a trend of delayed viral acquisition was found in vaccinees compared to controls, which reached statistical significance in animals with the TRIM-5α-resistant (TRIM-5α R) allele. Vaccinees were preferentially infected by an SIVsmE660 transmitted/founder virus carrying neutralization-resistant A/K mutations at residues 45 and 47 in Env, demonstrating a strong vaccine-induced sieve effect. In addition, the delay in virus acquisition directly correlated with SIVsmE660-specific neutralizing antibodies. The presence of mucosal V1V2 IgG binding antibodies correlated with a significantly decreased risk of virus acquisition in both TRIM-5α R and TRIM-5α-moderate/sensitive (TRIM-5α M/S) animals, although this vaccine effect was more prominent in animals with the TRIM-5α R allele. These data support the combined contribution of immune responses and genetic background to vaccine efficacy. Humoral responses targeting V2 and SIV-specific T cell responses correlated with viremia control. In conclusion, the combination of DNA and gp120 Env protein vaccine regimens using two different adjuvants induced durable and potent cellular and humoral responses contributing to a lower risk of infection by heterologous SIV challenge.IMPORTANCE An effective AIDS vaccine continues to be of paramount importance for the control of the pandemic, and it has been proven to be an elusive target. Vaccine efficacy trials and macaque challenge studies indicate that protection may be the result of combinations of many parameters. We show that a combination of DNA and protein vaccinations applied at the same time provides rapid and robust cellular and humoral immune responses and evidence for a reduced risk of infection. Vaccine-induced neutralizing antibodies and Env V2-specific antibodies at mucosal sites contribute to the delay of SIVsmE660 acquisition, and genetic makeup (TRIM-5α) affects the effectiveness of the vaccine. These data are important for the design of better vaccines and may also affect other vaccine platforms.
Keywords: A/K variant; ADCC; ADCD; ADNP; Ab glycosylation structures; DNA; HIV; QS21; SIVmac251; SIVsmE660; SIVsmE660 T/F; T cell responses; TLR4; TLR7; TRIM-5α; V2 responses; acquisition delay; adjuvant; binding antibody; correlate of viremia control; cyclic V2; humoral responses; immunization; linear peptide; mucosal responses; neutralizing antibody; protein; reduced risk of infection; repeated low-dose rectal challenge; rhesus macaque; scaffolded gp70-V1V2; systems serology; vaccination; vaccine; viremia control.
Copyright © 2018 Singh et al.
Figures



Similar articles
-
Adjuvanting a Simian Immunodeficiency Virus Vaccine with Toll-Like Receptor Ligands Encapsulated in Nanoparticles Induces Persistent Antibody Responses and Enhanced Protection in TRIM5α Restrictive Macaques.J Virol. 2017 Jan 31;91(4):e01844-16. doi: 10.1128/JVI.01844-16. Print 2017 Feb 15. J Virol. 2017. PMID: 27928002 Free PMC article.
-
Virus-Like Particles Displaying Trimeric Simian Immunodeficiency Virus (SIV) Envelope gp160 Enhance the Breadth of DNA/Modified Vaccinia Virus Ankara SIV Vaccine-Induced Antibody Responses in Rhesus Macaques.J Virol. 2016 Sep 12;90(19):8842-54. doi: 10.1128/JVI.01163-16. Print 2016 Oct 1. J Virol. 2016. PMID: 27466414 Free PMC article.
-
Vaccination with Vaxfectin(®) adjuvanted SIV DNA induces long-lasting humoral immune responses able to reduce SIVmac251 Viremia.Hum Vaccin Immunother. 2013 Oct;9(10):2069-80. doi: 10.4161/hv.25442. Epub 2013 Jul 2. Hum Vaccin Immunother. 2013. PMID: 23820294 Free PMC article.
-
Nonneutralizing functional antibodies: a new "old" paradigm for HIV vaccines.Clin Vaccine Immunol. 2014 Aug;21(8):1023-36. doi: 10.1128/CVI.00230-14. Epub 2014 Jun 11. Clin Vaccine Immunol. 2014. PMID: 24920599 Free PMC article. Review.
-
Mechanisms of protection against simian immunodeficiency virus infection.Vaccine. 2002 May 6;20(15):1985-7. doi: 10.1016/s0264-410x(02)00083-x. Vaccine. 2002. PMID: 11983259 Review.
Cited by
-
Direct intranodal tonsil vaccination with modified vaccinia Ankara vaccine protects macaques from highly pathogenic SIVmac251.Nat Commun. 2023 Mar 7;14(1):1264. doi: 10.1038/s41467-023-36907-0. Nat Commun. 2023. PMID: 36882405 Free PMC article.
-
Revisiting the Correlate of Reduced HIV Infection Risk in the Rv144 Vaccine Trial.J Virol. 2019 Aug 13;93(17):e00629-19. doi: 10.1128/JVI.00629-19. Print 2019 Sep 1. J Virol. 2019. PMID: 31189712 Free PMC article.
-
HIV Vaccine Development at a Crossroads: New B and T Cell Approaches.Vaccines (Basel). 2024 Sep 12;12(9):1043. doi: 10.3390/vaccines12091043. Vaccines (Basel). 2024. PMID: 39340073 Free PMC article. Review.
-
Search for antiviral functions of potentially protective antibodies against V2 region of HIV-1.Hum Vaccin Immunother. 2020 Sep 1;16(9):2033-2041. doi: 10.1080/21645515.2020.1787070. Epub 2020 Jul 23. Hum Vaccin Immunother. 2020. PMID: 32701369 Free PMC article.
-
Differential V2-directed antibody responses in non-human primates infected with SHIVs or immunized with diverse HIV vaccines.Nat Commun. 2022 Feb 16;13(1):903. doi: 10.1038/s41467-022-28450-1. Nat Commun. 2022. PMID: 35173151 Free PMC article.
References
-
- Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, Paris R, Premsri N, Namwat C, de Souza M, Adams E, Benenson M, Gurunathan S, Tartaglia J, McNeil JG, Francis DP, Stablein D, Birx DL, Chunsuttiwat S, Khamboonruang C, Thongcharoen P, Robb ML, Michael NL, Kunasol P, Kim JH. 2009. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N Engl J Med 361:2209–2220. doi:10.1056/NEJMoa0908492. - DOI - PubMed
-
- Zolla-Pazner S, deCamp AC, Cardozo T, Karasavvas N, Gottardo R, Williams C, Morris DE, Tomaras G, Rao M, Billings E, Berman P, Shen X, Andrews C, O'Connell RJ, Ngauy V, Nitayaphan S, de Souza M, Korber B, Koup R, Bailer RT, Mascola JR, Pinter A, Montefiori D, Haynes BF, Robb ML, Rerks-Ngarm S, Michael NL, Gilbert PB, Kim JH. 2013. Analysis of V2 antibody responses induced in vaccinees in the ALVAC/AIDSVAX HIV-1 vaccine efficacy trial. PLoS One 8:e53629. doi:10.1371/journal.pone.0053629. - DOI - PMC - PubMed
-
- Haynes BF, Gilbert PB, McElrath MJ, Zolla-Pazner S, Tomaras GD, Alam SM, Evans DT, Montefiori DC, Karnasuta C, Sutthent R, Liao HX, DeVico AL, Lewis GK, Williams C, Pinter A, Fong Y, Janes H, DeCamp A, Huang Y, Rao M, Billings E, Karasavvas N, Robb ML, Ngauy V, de Souza MS, Paris R, Ferrari G, Bailer RT, Soderberg KA, Andrews C, Berman PW, Frahm N, De Rosa SC, Alpert MD, Yates NL, Shen X, Koup RA, Pitisuttithum P, Kaewkungwal J, Nitayaphan S, Rerks-Ngarm S, Michael NL, Kim JH. 2012. Immune-correlates analysis of an HIV-1 vaccine efficacy trial. N Engl J Med 366:1275–1286. doi:10.1056/NEJMoa1113425. - DOI - PMC - PubMed
-
- Rolland M, Edlefsen PT, Larsen BB, Tovanabutra S, Sanders-Buell E, Hertz T, deCamp AC, Carrico C, Menis S, Magaret CA, Ahmed H, Juraska M, Chen L, Konopa P, Nariya S, Stoddard JN, Wong K, Zhao H, Deng W, Maust BS, Bose M, Howell S, Bates A, Lazzaro M, O'Sullivan A, Lei E, Bradfield A, Ibitamuno G, Assawadarachai V, O'Connell RJ, deSouza MS, Nitayaphan S, Rerks-Ngarm S, Robb ML, McLellan JS, Georgiev I, Kwong PD, Carlson JM, Michael NL, Schief WR, Gilbert PB, Mullins JI, Kim JH. 2012. Increased HIV-1 vaccine efficacy against viruses with genetic signatures in Env V2. Nature 490:417–420. doi:10.1038/nature11519. - DOI - PMC - PubMed
-
- Rosati M, von Gegerfelt A, Roth P, Alicea C, Valentin A, Robert-Guroff M, Venzon D, Montefiori DC, Markham P, Felber BK, Pavlakis GN. 2005. DNA vaccines expressing different forms of simian immunodeficiency virus antigens decrease viremia upon SIVmac251 challenge. J Virol 79:8480–8492. doi:10.1128/JVI.79.13.8480-8492.2005. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

