Prevention and treatment of bleomycin-induced pulmonary fibrosis with the lactate dehydrogenase inhibitor gossypol
- PMID: 29795645
- PMCID: PMC5967738
- DOI: 10.1371/journal.pone.0197936
Prevention and treatment of bleomycin-induced pulmonary fibrosis with the lactate dehydrogenase inhibitor gossypol
Abstract
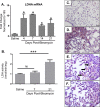
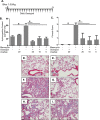
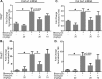
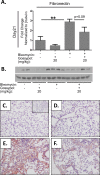
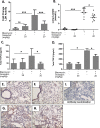
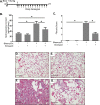
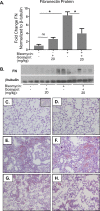
Pulmonary fibrosis is a chronic and irreversible scarring disease in the lung with poor prognosis. Few therapies are available; therefore it is critical to identify new therapeutic targets. Our lab has previously identified the enzyme lactate dehydrogenase-A (LDHA) as a potential therapeutic target in pulmonary fibrosis. We found increases in LDHA protein and its metabolic product, lactate, in patients with idiopathic pulmonary fibrosis (IPF). Importantly, we described lactate as a novel pro-fibrotic mediator by acidifying the extracellular space, and activating latent transforming growth factor beta (TGF-β1) in a pH-dependent manner. We propose a pro-fibrotic feed-forward loop by which LDHA produces lactate, lactate decreases pH in the extracellular space and activates TGF-β1 which can further perpetuate fibrotic signaling. Our previous work also demonstrates that the LDHA inhibitor gossypol inhibits TGF-β1-induced myofibroblast differentiation and collagen production in vitro. Here, we employed a mouse model of bleomycin-induced pulmonary fibrosis to test whether gossypol inhibits pulmonary fibrosis in vivo. We found that gossypol dose-dependently inhibits bleomycin-induced collagen accumulation and TGF-β1 activation in mouse lungs when treatment is started on the same day as bleomycin administration. Importantly, gossypol was also effective at treating collagen accumulation when delayed 7 days following bleomycin. Our results demonstrate that inhibition of LDHA with the inhibitor gossypol is effective at both preventing and treating bleomycin-induced pulmonary fibrosis, and suggests that LDHA may be a potential therapeutic target for pulmonary fibrosis.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Ley B, Collard HR, Talmadge E. King J (2011) Clinical Course and Prediction of Survival in Idiopathic Pulmonary Fibrosis. American Journal of Respiratory and Critical Care Medicine 183: 431–440. doi: 10.1164/rccm.201006-0894CI - DOI - PubMed
-
- Hinz B SH P, Thannickal V, Galli A, Bochaton-Piallat M, Gabbiani G. (2007) The myofibroblast: one function, multiple organs. Am J Pathol: 1807–1816. doi: 10.2353/ajpath.2007.070112 - DOI - PMC - PubMed
-
- Gabbiani G (2003) The myofibroblast in wound healing and fibrocontractive diseases. J Pathol: 500–503. doi: 10.1002/path.1427 - DOI - PubMed
-
- Baur PS Jr., Parks DH (1983) The myofibroblast anchoring strand—the fibronectin connection in wound healing and the possible loci of collagen fibril assembly. J Trauma 23: 853–862. - PubMed
-
- Poobalarahi F, Baicu CF, Bradshaw AD (2006) Cardiac myofibroblasts differentiated in 3D culture exhibit distinct changes in collagen I production, processing, and matrix deposition. Am J Physiol Heart Circ Physiol 291: H2924–2932. doi: 10.1152/ajpheart.00153.2006 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

